The Pediatric Clinical Trials Market was valued at USD 21.49 billion in 2023 and is expected to reach USD 28.63 billion by 2032, growing at a CAGR of 4.30% from 2024 to 2032.
The Pediatric Clinical Trials Market report offers a differentiated perspective through analysis of the rates of participation and enrollment, presenting a level of age-group and phase-specific segmentation to depict patient participation patterns. It includes a regional distribution breakdown, depicting major countries in pediatric research, as well as growth markets. The report assesses success rates and completion timeframes, noting influences on trial efficiency and length. Also, it discusses trends in funding and investments, with reports of government, private sector, and public-private partnership contributions. Finally, the report presents an overview of the regulatory landscape, mapping recent approvals and developments in pediatrics-focused drug development in major health authorities.
The U.S. Pediatric Clinical Trials Market size was USD 6.41 billion in 2023 and is expected to reach USD 8.34 billion by 2032, growing at a CAGR of 4.03% over the forecast period of 2024-2032.
The market is driven by increasing research efforts to develop safe and effective treatments for pediatric diseases, including rare genetic disorders and chronic conditions. Regulatory initiatives promoting pediatric drug development, along with advancements in clinical trial methodologies, are supporting market growth. Growing awareness about the need for age-specific drug testing and rising funding from government and private organizations are also contributing to the expansion of pediatric clinical trials in the U.S.
Drivers
Rising Burden of Pediatric Diseases and Unmet Medical Needs are propelling the market growth.
Growth in the incidence of child diseases, such as rare genetic disorders, congenital diseases, and children's cancer, is propelling demand for clinical trials specific to children. According to the American Cancer Society, almost 10,000 children under 15 years are diagnosed with cancer every year in the U.S., making ongoing research for improved treatment necessary. Likewise, the World Health Organization (WHO) estimates that more than 200 million children around the world live with chronic disease, most of whom have inadequate therapeutic options available. Recent regulation efforts, such as the U.S. FDA's Pediatric Research Equity Act (PREA) and the European Medicines Agency (EMA) Pediatric Regulation, require pharmaceutical corporations to carry out pediatric-specific clinical trials before approving adult medications for children. These factors are driving sponsors and CROs to grow pediatric clinical trial pipelines to meet such unmet needs.
Government Incentives and Regulatory Support for Pediatric Trials are accelerating the market to grow.
Governments and regulatory authorities across the globe are intensifying policies to promote pediatric clinical research. Within the U.S., the Best Pharmaceuticals for Children Act (BPCA) and the PREA offer incentives such as extended exclusivity for drug makers performing pediatric trials. The FDA's Rare Pediatric Disease Priority Review Voucher Program also encourages firms that are working on therapies for rare childhood diseases. Likewise, in 2023, the European Medicines Agency (EMA) implemented increased funding for pediatric trial networks, simplifying cooperation between researchers and pharmaceutical companies. These initiatives compensate for the high expenses and intricacies involved in pediatric research. Moreover, the initiation of international pediatric trial networks, like the International Neonatal Consortium (INC) and the Pediatric Trials Network (PTN), is enhancing infrastructure and patient enrollment. This regulatory and financial support is largely responsible for the increased number of pediatric clinical trials.
Restraint
Clinical trials in children are an enormous ethical and recruitment challenge, constraining market growth.
Pediatric trials involve rigorous ethical issues since children are not able to give informed consent and require parental or guardian permission. This most often results in low enrollment rates, which causes trial completion to be delayed and drug development to be protracted. In a study released in Pediatrics, more than 50% of pediatric clinical trials have faced recruitment issues, resulting in termination or reformulation of the trials. Furthermore, numerous parents are reluctant to enroll their children because of the fear of possible risks, side effects, and long-term effects. Agencies like the FDA and EMA have strict guidelines for pediatric trials, adding complexity to study design and conduct. All these elements combined raise the cost of trials, extend development times, and reduce the number of pediatric-specific treatments available, creating a primary challenge for pharmaceutical firms and research institutions.
Opportunities
Expansion of Pediatric Rare Disease Trials presents a significant opportunity for the market.
The growing emphasis on rare pediatric diseases offers tremendous growth prospects for the pediatric clinical trials market. Such diseases, with small patient populations, have limited or no effective treatments, thereby offering a high unmet medical need. Consequently, there is an emerging trend towards targeted therapies and novel treatments, resulting in increased trials for pediatric rare diseases. For example, programs such as the U.S. Orphan Drug Act and European orphan drug legislation offer incentives, including tax credits and market exclusivity, to stimulate the development of treatments for pediatric rare diseases. This regulatory encouragement is increasing investment in clinical trials and research for pediatric medicines, promoting partnerships among pharmaceutical firms, research institutions, and healthcare providers to speed drug development for these vulnerable populations.
Challenges
One of the foremost challenges in the pediatric clinical trials industry is effective navigation through the intricate regulatory system.
Clinical trials involving pediatric subjects are overseen by very strict ethical codes and regulatory provisions intended to uphold the safety and well-being of participants who are children. Bodies like the FDA, EMA, and ICH have formal processes of approvals that are elaborate and time-consuming, thereby dragging clinical trial initiations and execution behind. In addition, regional variations in regulations complicate multinational studies' ability to be both consistent and compliant. These issues result in greater costs, delays, and decreased access to necessary treatments for children. Pharmaceutical companies and researchers have to navigate efficiency, ethics, and regulatory compliance, a balancing act that can slow the rate of pediatric clinical research.
By Phase
The Phase III segment dominated the pediatric clinical trials market with a 55.24% market share in 2023 because it plays a key role in proving the safety and efficacy of novel treatments before regulatory approval. These trials involve a larger population of patients and are necessary to gain approval from such agencies as the FDA and EMA. Phase III pediatric trials are usually required by regulatory bodies, especially for diseases such as rare genetic disorders, oncology, and infectious diseases, to ensure that treatments are of high safety standards before they hit the market. Further, more government and industry investment in late-stage pediatric trials, as well as more pediatric drug development activity, helped drive this segment's leadership.
The Phase I segment is anticipated to exhibit the fastest growth in the forecast period based on the increasing emphasis on early-stage drug discovery and precision medicine for pediatric illness. Gene therapy, biologics, and orphan drugs have improved, leading to more early-phase trials focused on rare and complex pediatric diseases. Regulatory incentives, including priority review vouchers for rare pediatric diseases, are also pushing more investments into Phase I trials. Furthermore, the growth in pediatric oncology research and the requirement for age-specific formulations are encouraging pharmaceutical companies to launch more first-in-human trials in children, driving growth in this segment.
By Study Design
The Treatment Studies segment dominated the pediatric clinical trials market with a 68.28% market share in 2023 as a result of growing demand for efficient treatments of diseases in children, such as rare genetic diseases, oncology, and infectious diseases. The trials are designed to test the safety and effectiveness of new drugs, biologics, and new treatment modalities, which render them crucial for regulatory clearance and clinical innovation. Moreover, intense government efforts, funding from bodies such as the National Institutes of Health (NIH) and the European Medicines Agency (EMA), and pharma investments in drug development for the pediatric population have fueled the rise of treatment-oriented clinical trials. Increased focus on personalized medicine and the increasing pipeline of pediatric-specific formulations have also supported the dominance of this category, with scientists looking to create age-specific and targeted drugs for children.
By Indication
The Oncology segment dominated the pediatric clinical trials market with 43.20% market share in 2023 because of the increasing rate of cancers among children, such as leukemia, brain cancer, and neuroblastoma, that demand ongoing research for better treatment. Pediatric oncology trials are well funded by international healthcare organizations, governments, and private institutions to increase survival rates and reduce the long-term toxicities among children. The creation of targeted therapies, immunotherapies, and CAR-T cell therapies has further boosted clinical trial activity. Moreover, regulatory incentives like the FDA's Rare Pediatric Disease Priority Review Voucher Program incentivize pharmaceutical firms to invest in pediatric oncology research, further cementing the segment's dominance.
Regional Analysis
North America dominated the pediatric clinical trials market with a 41.26% market share in 2023 because of its well-developed healthcare infrastructure, high research funding, and regulatory assistance. The region is inhabited by some of the world's most prominent pharmaceutical companies, research centers, and hospitals, which undertake a high volume of pediatric clinical trials. The U.S. Food and Drug Administration (FDA) and other regulatory authorities in North America offer a supportive climate with programs such as pediatric exclusivity and orphan drug designations, which stimulate research into pediatric illnesses. The high rate of healthcare expenditure, combined with a strong system for patient recruitment and retention, has also established North America as a center for sophisticated pediatric clinical trials, further solidifying its leadership.
Asia Pacific is the fastest-growing region in the Pediatric Clinical Trials market with 4.88% CAGR. This high growth is attributable to several factors such as rising healthcare investments, growing healthcare infrastructure, and growing numbers of clinical trial outsourcing activities to nations like India, China, and South Korea. Moreover, the high pediatric population of the region and increasing demand for treatments of common and uncommon diseases provide immense opportunities for clinical trials. The regulatory authorities in the region are also changing to make clinical trial approvals more streamlined, and hence, the region is a favorable destination for clinical research. The growing economic growth, with rising demand for enhanced healthcare facilities, underlies the growth of clinical trial operations, setting up Asia Pacific to achieve fast development in the following years.
Pfizer Inc. (Prevnar 13, Ibrance)
Merck & Co., Inc. (Gardasil, Keytruda)
Novartis (Kymriah, Aimovig)
Johnson & Johnson (Janssen Pediatric Vaccine, Remicade)
AstraZeneca (Synagis, Imfinzi)
GlaxoSmithKline (GSK) (Bexsero, Synflorix)
Roche Pharmaceuticals (Actemra, Herceptin)
Eli Lilly and Company (Mounjaro, Verzenio)
Sanofi (Dengvaxia, Dupixent)
Bayer AG (Kovaltry, Adempas)
Bristol-Myers Squibb (Opdivo, Yervoy)
AbbVie Inc. (Humira, Rinvoq)
Novo Nordisk (Norditropin, Ozempic)
Regeneron Pharmaceuticals (Eylea, Dupixent)
Amgen Inc. (Enbrel, Xgeva)
GlaxoSmithKline (GSK) (Fluarix, Bexsero)
Baxter International Inc. (HyQvia, Adynovate)
UCB S.A. (Briviact, Vimpat)
Sobi AB (Kineret, Elocta)
Teva Pharmaceutical Industries (Copaxone, Austedo)
Suppliers (These suppliers provide essential services in clinical trial management, including regulatory support, clinical monitoring, data management, and specialized pediatric research, ensuring that clinical trials for pediatric products are conducted effectively and in compliance with health regulations.) in Pediatric Clinical Trials Market
Lonza Group
WuXi AppTec
Parexel International
Celerion
Covance (Labcorp Drug Development)
Charles River Laboratories
PPD (Pharmaceutical Product Development)
Medpace
Syneos Health
ICON plc
In June 2024, Merck, also known as MSD outside of the U.S. and Canada, released positive topline results from its Phase 2b/3 clinical trial (MK-1654-004) assessing clesrovimab (MK-1654), an investigational prophylactic monoclonal antibody for the prevention of RSV disease in infants. The trial achieved its main safety and efficacy endpoints, such as a decrease in medically attended lower respiratory illness (MALRI) due to RSV through Day 150. Merck will provide the detailed results at an upcoming scientific meeting and submit these data to worldwide regulatory authorities.
In March 2024, Novartis shared new evidence from the Phase IIIb open-label SMART trial, which assessed the safety and effectiveness of its approved gene therapy, Zolgensma (onasemnogene abeparvovec), in older and heavier children with spinal muscular atrophy (SMA).
On February 9, 2024, Johnson & Johnson announced that the U.S. Food and Drug Administration (FDA) granted Breakthrough Therapy Designation (BTD) for nipocalimab for the treatment of alloimmunized pregnant women at increased risk of life-threatening hemolytic disease of the fetus and newborn (HDFN). Nipocalimab is the only treatment reported to be in development for this serious and rare condition, which happens when the blood types of the pregnant woman and fetus are incompatible and can cause life-threatening anemia in the fetus or infant.
| Report Attributes | Details |
|---|---|
| Market Size in 2023 | US$ 21.49 Billion |
| Market Size by 2032 | US$ 28.63 Billion |
| CAGR | CAGR of 4.30% from 2024 to 2032 |
| Base Year | 2023 |
| Forecast Period | 2024-2032 |
| Historical Data | 2020-2022 |
| Report Scope & Coverage | Market Size, Segments Analysis, Competitive Landscape, Regional Analysis, DROC & SWOT Analysis, Forecast Outlook |
| Key Segments | • By Phase (Phase I, Phase II, Phase III, Phase IV) • By Study Design (Treatment Studies, Observational Studies) • By Indication (Infectious Diseases, Oncology, Autoimmune/Inflammation, Respiratory Disorders, Mental Health Disorders, Others) |
| Regional Analysis/Coverage | North America (US, Canada, Mexico), Europe (Eastern Europe [Poland, Romania, Hungary, Turkey, Rest of Eastern Europe] Western Europe] Germany, France, UK, Italy, Spain, Netherlands, Switzerland, Austria, Rest of Western Europe]), Asia Pacific (China, India, Japan, South Korea, Vietnam, Singapore, Australia, Rest of Asia Pacific), Middle East & Africa (Middle East [UAE, Egypt, Saudi Arabia, Qatar, Rest of Middle East], Africa [Nigeria, South Africa, Rest of Africa], Latin America (Brazil, Argentina, Colombia, Rest of Latin America) |
| Company Profiles | Pfizer Inc., Merck & Co., Inc., Novartis, Johnson & Johnson, AstraZeneca, GlaxoSmithKline (GSK), Roche Pharmaceuticals, Eli Lilly and Company, Sanofi, Bayer AG, Bristol-Myers Squibb, AbbVie Inc., Novo Nordisk, Regeneron Pharmaceuticals, Amgen Inc., Baxter International Inc., UCB S.A., Sobi AB, Teva Pharmaceutical Industries, and other players. |
Ans: The Pediatric Clinical Trials Market is expected to grow at a CAGR of 4.30% from 2024-2032.
Ans: The Pediatric Clinical Trials Market was USD 21.49 billion in 2023 and is expected to reach USD 28.63 billion by 2032.
Ans: Government Incentives and Regulatory Support for Pediatric Trials are accelerating the market to grow.
Ans: The “Phase III” segment dominated the Pediatric Clinical Trials Market.
Ans: North America dominated the Pediatric Clinical Trials Market in 2023.
Table of Contents:
1. Introduction
1.1 Market Definition
1.2 Scope (Inclusion and Exclusions)
1.3 Research Assumptions
2. Executive Summary
2.1 Market Overview
2.2 Regional Synopsis
2.3 Competitive Summary
3. Research Methodology
3.1 Top-Down Approach
3.2 Bottom-up Approach
3.3. Data Validation
3.4 Primary Interviews
4. Market Dynamics Impact Analysis
4.1 Market Driving Factors Analysis
4.1.1 Drivers
4.1.2 Restraints
4.1.3 Opportunities
4.1.4 Challenges
4.2 PESTLE Analysis
4.3 Porter’s Five Forces Model
5. Statistical Insights and Trends Reporting
5.1 Enrollment and Participation Rates in Pediatric Clinical Trials (2023)
5.2 Success Rates and Completion Timelines of Pediatric Clinical Trials (2020-2032)
5.3 Regional Distribution of Pediatric Clinical Trials (2023)
5.4 Funding and Investment in Pediatric Clinical Research (2023)
5.5 Regulatory Approvals and Drug Development Trends for Pediatric Treatments (2023)
6. Competitive Landscape
6.1 List of Major Companies, By Region
6.2 Market Share Analysis, By Region
6.3 Product Benchmarking
6.3.1 Product specifications and features
6.3.2 Pricing
6.4 Strategic Initiatives
6.4.1 Marketing and promotional activities
6.4.2 Distribution and supply chain strategies
6.4.3 Expansion plans and new product launches
6.4.4 Strategic partnerships and collaborations
6.5 Technological Advancements
6.6 Market Positioning and Branding
7. Pediatric Clinical Trials Market Segmentation, By Phase
7.1 Chapter Overview
7.2 Phase I
7.2.1 Phase I Market Trends Analysis (2020-2032)
7.2.2 Phase I Market Size Estimates and Forecasts to 2032 (USD Billion)
7.3 Phase II
7.3.1 Phase II Market Trends Analysis (2020-2032)
7.3.2 Phase II Market Size Estimates and Forecasts to 2032 (USD Billion)
7.4 Phase III
7.4.1 Phase III Market Trends Analysis (2020-2032)
7.4.2 Phase III Market Size Estimates and Forecasts to 2032 (USD Billion)
7.5 Phase IV
7.5.1 Phase IV Market Trends Analysis (2020-2032)
7.5.2 Phase IV Market Size Estimates and Forecasts to 2032 (USD Billion)
8. Pediatric Clinical Trials Market Segmentation, By Study Design
8.1 Chapter Overview
8.2 Treatment Studies
8.2.1 Treatment Studies Market Trends Analysis (2020-2032)
8.2.2 Treatment Studies Market Size Estimates and Forecasts to 2032 (USD Billion)
8.3 Observational Studies
8.3.1 Observational Studies Market Trends Analysis (2020-2032)
8.3.2 Observational Studies Market Size Estimates and Forecasts to 2032 (USD Billion)
9. Pediatric Clinical Trials Market Segmentation, By Indication
9.1 Chapter Overview
9.2 Infectious Diseases
9.2.1 Infectious Diseases Market Trends Analysis (2020-2032)
9.2.2 Infectious Diseases Market Size Estimates and Forecasts to 2032 (USD Billion)
9.3 Oncology
9.3.1 Oncology Market Trends Analysis (2020-2032)
9.3.2 Oncology Market Size Estimates and Forecasts to 2032 (USD Billion)
9.4 Autoimmune/inflammation
9.4.1 Autoimmune/inflammation Market Trends Analysis (2020-2032)
9.4.2 Autoimmune/inflammation Market Size Estimates and Forecasts to 2032 (USD Billion)
9.5 Respiratory Disorders
9.5.1 Respiratory Disorders Market Trends Analysis (2020-2032)
9.5.2 Respiratory Disorders Market Size Estimates and Forecasts to 2032 (USD Billion)
9.6 Mental Health Disorders
9.6.1 Mental Health Disorders Market Trends Analysis (2020-2032)
9.6.2 Mental Health Disorders Market Size Estimates and Forecasts to 2032 (USD Billion)
9.6 Others
9.6.1 Others Market Trends Analysis (2020-2032)
9.6.2 Others Market Size Estimates and Forecasts to 2032 (USD Billion)
10. Regional Analysis
10.1 Chapter Overview
10.2 North America
10.2.1 Trends Analysis
10.2.2 North America Pediatric Clinical Trials Market Estimates and Forecasts, by Country (2020-2032) (USD Billion)
10.2.3 North America Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.2.4 North America Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.2.5 North America Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.2.6 USA
10.2.6.1 USA Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.2.6.2 USA Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.2.6.3 USA Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.2.7 Canada
10.2.7.1 Canada Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.2.7.2 Canada Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.2.7.3 Canada Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.2.8 Mexico
10.2.8.1 Mexico Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.2.8.2 Mexico Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.2.8.3 Mexico Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.3 Europe
10.3.1 Eastern Europe
10.3.1.1 Trends Analysis
10.3.1.2 Eastern Europe Pediatric Clinical Trials Market Estimates and Forecasts, by Country (2020-2032) (USD Billion)
10.3.1.3 Eastern Europe Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.1.4 Eastern Europe Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.3.1.5 Eastern Europe Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.3.1.6 Poland
10.3.1.6.1 Poland Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.1.6.2 Poland Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.3.1.6.3 Poland Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.3.1.7 Romania
10.3.1.7.1 Romania Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.1.7.2 Romania Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.3.1.7.3 Romania Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.3.1.8 Hungary
10.3.1.8.1 Hungary Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.1.8.2 Hungary Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.3.1.8.3 Hungary Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.3.1.9 Turkey
10.3.1.9.1 Turkey Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.1.9.2 Turkey Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.3.1.9.3 Turkey Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.3.1.10 Rest of Eastern Europe
10.3.1.10.1 Rest of Eastern Europe Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.1.10.2 Rest of Eastern Europe Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.3.1.10.3 Rest of Eastern Europe Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.3.2 Western Europe
10.3.2.1 Trends Analysis
10.3.2.2 Western Europe Pediatric Clinical Trials Market Estimates and Forecasts, by Country (2020-2032) (USD Billion)
10.3.2.3 Western Europe Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.2.4 Western Europe Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.3.2.5 Western Europe Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.3.2.6 Germany
10.3.2.6.1 Germany Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.2.6.2 Germany Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.3.2.6.3 Germany Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.3.2.7 France
10.3.2.7.1 France Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.2.7.2 France Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.3.2.7.3 France Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.3.2.8 UK
10.3.2.8.1 UK Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.2.8.2 UK Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.3.2.8.3 UK Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.3.2.9 Italy
10.3.2.9.1 Italy Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.2.9.2 Italy Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.3.2.9.3 Italy Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.3.2.10 Spain
10.3.2.10.1 Spain Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.2.10.2 Spain Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.3.2.10.3 Spain Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.3.2.11 Netherlands
10.3.2.11.1 Netherlands Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.2.11.2 Netherlands Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.3.2.11.3 Netherlands Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.3.2.12 Switzerland
10.3.2.12.1 Switzerland Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.2.12.2 Switzerland Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.3.2.12.3 Switzerland Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.3.2.13 Austria
10.3.2.13.1 Austria Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.2.13.2 Austria Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.3.2.13.3 Austria Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.3.2.14 Rest of Western Europe
10.3.2.14.1 Rest of Western Europe Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.2.14.2 Rest of Western Europe Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.3.2.14.3 Rest of Western Europe Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.4 Asia Pacific
10.4.1 Trends Analysis
10.4.2 Asia Pacific Pediatric Clinical Trials Market Estimates and Forecasts, by Country (2020-2032) (USD Billion)
10.4.3 Asia Pacific Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.4.4 Asia Pacific Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.4.5 Asia Pacific Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.4.6 China
10.4.6.1 China Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.4.6.2 China Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.4.6.3 China Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.4.7 India
10.4.7.1 India Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.4.7.2 India Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.4.7.3 India Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.4.8 Japan
10.4.8.1 Japan Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.4.8.2 Japan Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.4.8.3 Japan Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.4.9 South Korea
10.4.9.1 South Korea Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.4.9.2 South Korea Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.4.9.3 South Korea Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.4.10 Vietnam
10.4.10.1 Vietnam Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.4.10.2 Vietnam Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.4.10.3 Vietnam Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.4.11 Singapore
10.4.11.1 Singapore Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.4.11.2 Singapore Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.4.11.3 Singapore Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.4.12 Australia
10.4.12.1 Australia Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.4.12.2 Australia Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.4.12.3 Australia Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.4.13 Rest of Asia Pacific
10.4.13.1 Rest of Asia Pacific Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.4.13.2 Rest of Asia Pacific Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.4.13.3 Rest of Asia Pacific Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.5 Middle East and Africa
10.5.1 Middle East
10.5.1.1 Trends Analysis
10.5.1.2 Middle East Pediatric Clinical Trials Market Estimates and Forecasts, by Country (2020-2032) (USD Billion)
10.5.1.3 Middle East Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.5.1.4 Middle East Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.5.1.5 Middle East Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.5.1.6 UAE
10.5.1.6.1 UAE Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.5.1.6.2 UAE Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.5.1.6.3 UAE Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.5.1.7 Egypt
10.5.1.7.1 Egypt Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.5.1.7.2 Egypt Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.5.1.7.3 Egypt Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.5.1.8 Saudi Arabia
10.5.1.8.1 Saudi Arabia Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.5.1.8.2 Saudi Arabia Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.5.1.8.3 Saudi Arabia Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.5.1.9 Qatar
10.5.1.9.1 Qatar Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.5.1.9.2 Qatar Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.5.1.9.3 Qatar Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.5.1.10 Rest of Middle East
10.5.1.10.1 Rest of Middle East Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.5.1.10.2 Rest of Middle East Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.5.1.10.3 Rest of Middle East Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.5.2 Africa
10.5.2.1 Trends Analysis
10.5.2.2 Africa Pediatric Clinical Trials Market Estimates and Forecasts, by Country (2020-2032) (USD Billion)
10.5.2.3 Africa Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.5.2.4 Africa Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.5.2.5 Africa Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.5.2.6 South Africa
10.5.2.6.1 South Africa Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.5.2.6.2 South Africa Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.5.2.6.3 South Africa Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.5.2.7 Nigeria
10.5.2.7.1 Nigeria Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.5.2.7.2 Nigeria Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.5.2.7.3 Nigeria Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.5.2.8 Rest of Africa
10.5.2.8.1 Rest of Africa Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.5.2.8.2 Rest of Africa Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.5.2.8.3 Rest of Africa Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.6 Latin America
10.6.1 Trends Analysis
10.6.2 Latin America Pediatric Clinical Trials Market Estimates and Forecasts, by Country (2020-2032) (USD Billion)
10.6.3 Latin America Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.6.4 Latin America Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.6.5 Latin America Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.6.6 Brazil
10.6.6.1 Brazil Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.6.6.2 Brazil Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.6.6.3 Brazil Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.6.7 Argentina
10.6.7.1 Argentina Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.6.7.2 Argentina Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.6.7.3 Argentina Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.6.8 Colombia
10.6.8.1 Colombia Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.6.8.2 Colombia Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.6.8.3 Colombia Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
10.6.9 Rest of Latin America
10.6.9.1 Rest of Latin America Pediatric Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.6.9.2 Rest of Latin America Pediatric Clinical Trials Market Estimates and Forecasts, by Study Design (2020-2032) (USD Billion)
10.6.9.3 Rest of Latin America Pediatric Clinical Trials Market Estimates and Forecasts, by Indication (2020-2032) (USD Billion)
11. Company Profiles
11.1 Pfizer Inc.
11.1.1 Company Overview
11.1.2 Financial
11.1.3 Products/ Services Offered
11.1.4 SWOT Analysis
11.2 Merck & Co., Inc.
11.2.1 Company Overview
11.2.2 Financial
11.2.3 Products/ Services Offered
11.2.4 SWOT Analysis
11.3 Novartis
11.3.1 Company Overview
11.3.2 Financial
11.3.3 Products/ Services Offered
11.3.4 SWOT Analysis
11.4 Johnson & Johnson
11.4.1 Company Overview
11.4.2 Financial
11.4.3 Products/ Services Offered
11.4.4 SWOT Analysis
11.5 AstraZeneca
11.5.1 Company Overview
11.5.2 Financial
11.5.3 Products/ Services Offered
11.5.4 SWOT Analysis
11.6 GlaxoSmithKline
11.6.1 Company Overview
11.6.2 Financial
11.6.3 Products/ Services Offered
11.6.4 SWOT Analysis
11.7 Roche Pharmaceuticals
11.7.1 Company Overview
11.7.2 Financial
11.7.3 Products/ Services Offered
11.7.4 SWOT Analysis
11.8 Eli Lilly and Company
11.8.1 Company Overview
11.8.2 Financial
11.8.3 Products/ Services Offered
11.8.4 SWOT Analysis
11.9 Sanofi
11.9.1 Company Overview
11.9.2 Financial
11.9.3 Products/ Services Offered
11.9.4 SWOT Analysis
11.10 Bayer AG
11.10.1 Company Overview
11.10.2 Financial
11.10.3 Products/ Services Offered
11.10.4 SWOT Analysis
12. Use Cases and Best Practices
13. Conclusion
An accurate research report requires proper strategizing as well as implementation. There are multiple factors involved in the completion of good and accurate research report and selecting the best methodology to compete the research is the toughest part. Since the research reports we provide play a crucial role in any company’s decision-making process, therefore we at SNS Insider always believe that we should choose the best method which gives us results closer to reality. This allows us to reach at a stage wherein we can provide our clients best and accurate investment to output ratio.
Each report that we prepare takes a timeframe of 350-400 business hours for production. Starting from the selection of titles through a couple of in-depth brain storming session to the final QC process before uploading our titles on our website we dedicate around 350 working hours. The titles are selected based on their current market cap and the foreseen CAGR and growth.
The 5 steps process:
Step 1: Secondary Research:
Secondary Research or Desk Research is as the name suggests is a research process wherein, we collect data through the readily available information. In this process we use various paid and unpaid databases which our team has access to and gather data through the same. This includes examining of listed companies’ annual reports, Journals, SEC filling etc. Apart from this our team has access to various associations across the globe across different industries. Lastly, we have exchange relationships with various university as well as individual libraries.
Step 2: Primary Research
When we talk about primary research, it is a type of study in which the researchers collect relevant data samples directly, rather than relying on previously collected data. This type of research is focused on gaining content specific facts that can be sued to solve specific problems. Since the collected data is fresh and first hand therefore it makes the study more accurate and genuine.
We at SNS Insider have divided Primary Research into 2 parts.
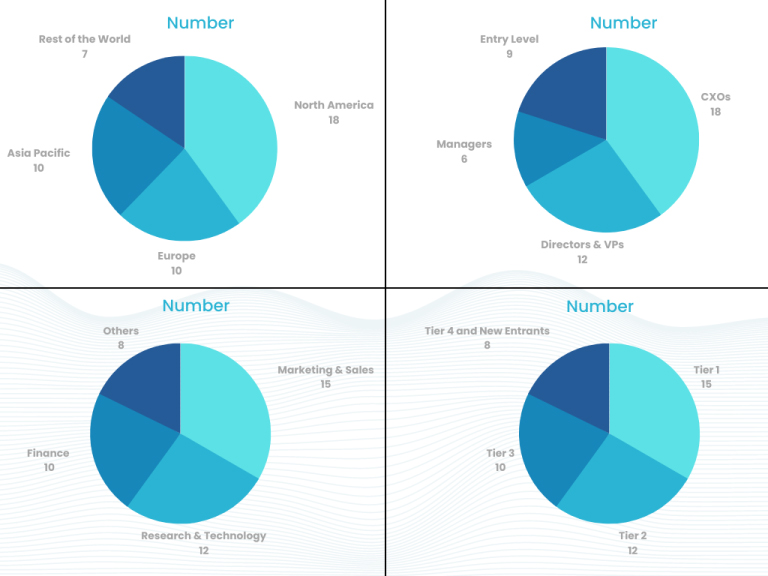
Part 1 wherein we interview the KOLs of major players as well as the upcoming ones across various geographic regions. This allows us to have their view over the market scenario and acts as an important tool to come closer to the accurate market numbers. As many as 45 paid and unpaid primary interviews are taken from both the demand and supply side of the industry to make sure we land at an accurate judgement and analysis of the market.
This step involves the triangulation of data wherein our team analyses the interview transcripts, online survey responses and observation of on filed participants. The below mentioned chart should give a better understanding of the part 1 of the primary interview.
Part 2: In this part of primary research the data collected via secondary research and the part 1 of the primary research is validated with the interviews from individual consultants and subject matter experts.
Consultants are those set of people who have at least 12 years of experience and expertise within the industry whereas Subject Matter Experts are those with at least 15 years of experience behind their back within the same space. The data with the help of two main processes i.e., FGDs (Focused Group Discussions) and IDs (Individual Discussions). This gives us a 3rd party nonbiased primary view of the market scenario making it a more dependable one while collation of the data pointers.
Step 3: Data Bank Validation
Once all the information is collected via primary and secondary sources, we run that information for data validation. At our intelligence centre our research heads track a lot of information related to the market which includes the quarterly reports, the daily stock prices, and other relevant information. Our data bank server gets updated every fortnight and that is how the information which we collected using our primary and secondary information is revalidated in real time.

Step 4: QA/QC Process
After all the data collection and validation our team does a final level of quality check and quality assurance to get rid of any unwanted or undesired mistakes. This might include but not limited to getting rid of the any typos, duplication of numbers or missing of any important information. The people involved in this process include technical content writers, research heads and graphics people. Once this process is completed the title gets uploader on our platform for our clients to read it.
Step 5: Final QC/QA Process:
This is the last process and comes when the client has ordered the study. In this process a final QA/QC is done before the study is emailed to the client. Since we believe in giving our clients a good experience of our research studies, therefore, to make sure that we do not lack at our end in any way humanly possible we do a final round of quality check and then dispatch the study to the client.
Pediatric Clinical Trials Market
Key Segments:
By Phase
Phase I
Phase II
Phase III
Phase IV
By Study Design
Treatment Studies
Observational Studies
By Indication
Infectious Diseases
Oncology
Autoimmune/inflammation
Respiratory Disorders
Mental Health Disorders
Others
Request for Segment Customization as per your Business Requirement: Segment Customization Request
Regional Coverage:
North America
US
Canada
Mexico
Europe
Eastern Europe
Poland
Romania
Hungary
Turkey
Rest of Eastern Europe
Western Europe
Germany
France
UK
Italy
Spain
Netherlands
Switzerland
Austria
Rest of Western Europe
Asia Pacific
China
India
Japan
South Korea
Vietnam
Singapore
Australia
Rest of Asia Pacific
Middle East & Africa
Middle East
UAE
Egypt
Saudi Arabia
Qatar
Rest of Middle East
Africa
Nigeria
South Africa
Rest of Africa
Latin America
Brazil
Argentina
Colombia
Rest of Latin America
Request for Country Level Research Report: Country Level Customization Request
Available Customization
With the given market data, SNS Insider offers customization as per the company’s specific needs. The following customization options are available for the report:
Detailed Volume Analysis
Criss-Cross segment analysis (e.g. Product X Application)
Competitive Product Benchmarking
Geographic Analysis
Additional countries in any of the regions
Customized Data Representation
Detailed analysis and profiling of additional market players
The Behavioral Rehabilitation Market was valued at USD 456.6 Billion in 2023 and is expected to reach USD 799.4 Billion by 2032, growing at a CAGR of 6.4% over the forecast period 2024-2032.
The Minimal Residual Disease Testing Market was valued at USD 2.16 billion in 2023 and is expected to reach USD 5.74 billion by 2032, growing at a CAGR of 11.50% over the forecast period of 2024-2032.
The Ferritin Testing Market Size was valued at USD 840.40 million in 2023 and is expected to reach USD 1684.34 million by 2032 and grow at a CAGR of 8.05% over the forecast period 2024-2032.
Molecular Imaging Market Size was valued at USD 8.47 billion in 2023 and is expected to reach USD 12.5 billion by 2032, growing at 4.5% CAGR from 2024-2032.
The Probe Reprocessing Market was valued at USD 752.42 million in 2023 and is expected to reach USD 1985.18 million by 2032, growing at a CAGR of 11.27% from 2024 to 2032.
The In Vitro Fertilization (IVF) Market was valued USD 18.82 billion in 2023, anticipated to reach USD 35.83 billion by 2032 with a compound annual growth rate at 7.47% during the forecast period 2024-2032.
Hi! Click one of our member below to chat on Phone