
Get More Information on Neonatal Respiratory Care Devices Market - Request Sample Report
The Neonatal Respiratory Care Devices Market size was valued at USD 1.82 billion In 2023 & is estimated to reach USD 3.10 billion by 2032 and increase at a compound annual growth rate of 6.1% between 2024 and 2032.
The Neonatal Respiratory Care Devices Market refers to the market for medical devices that are specifically designed to provide respiratory support and care to newborn infants, particularly those who are born prematurely or with respiratory issues. These devices are crucial for managing various respiratory conditions and ensuring proper oxygenation and ventilation in neonates, as their underdeveloped lungs and respiratory systems can pose significant challenges.
Neonatal respiratory care devices include a range of equipment designed to address different aspects of respiratory support, such as Ventilators are machines that assist or replace the natural breathing process by delivering controlled amounts of air or oxygen to the baby's lungs. Ventilators are used to treat conditions like respiratory distress syndrome (RDS) and help prevent lung damage. CPAP (Continuous Positive Airway Pressure) Devices deliver a continuous flow of air or oxygen to a baby's airways, helping to keep the airways open and improving oxygenation. CPAP is often used for conditions like RDS or to treat apnea of prematurity (brief pauses in breathing). High-Flow Nasal Cannula (HFNC) delivers humidified, high-flow oxygen through small prongs placed in the baby's nose. It provides oxygenation while also supporting respiratory effort and maintaining positive airway pressure. Nasal CPAP is Similar to traditional CPAP, this method uses prongs placed in the baby's nose to deliver a continuous flow of air or oxygen to help maintain open airways. Pulse Oximeters devices measure the oxygen saturation level in a baby's blood by attaching a small probe to the baby's skin.
They provide real-time monitoring of the baby's oxygen levels. Respiratory Monitors track various parameters related to the baby's respiratory function, such as respiratory rate, effort, and patterns. They help healthcare providers identify any abnormalities and adjust treatment accordingly. Surfactant Administration Devices is a substance that helps the baby's lungs inflate properly. Devices for administering surfactants can be used to treat respiratory distress in premature infants. The neonatal respiratory care devices market has grown significantly over the years due to advancements in medical technology, increased awareness of neonatal healthcare needs, and a greater emphasis on improving survival rates and reducing complications in premature and critically ill newborns. These devices play a crucial role in providing life-saving support to neonates with respiratory challenges, ensuring their proper development and growth. Neonatal Respiratory care devices are used in the diagnosis, monitoring, and treatment of respiratory disorders such as COPD, asthma, TB, and pneumonia. These devices improve the management of patients with acute and chronic respiratory illnesses. The study breaks down the respiratory care devices market by Product Type category, end user, and geography.
DRIVERS:
The incidence of premature births has been increasing due to various factors such as maternal age, multiple pregnancies, and medical conditions.
Rising Awareness and Better Healthcare Infrastructure is the driver of the Neonatal Respiratory Care Devices Market.
Increasing awareness among healthcare providers and parents about the importance of neonatal care, coupled with improved healthcare infrastructure in many regions, has led to a higher demand for quality respiratory care devices for newborns.
RESTRAIN:
Neonatal respiratory care devices can be expensive to develop, manufacture, and purchase.
The stringent Regulatory Approval Process restrains the Neonatal Respiratory Care Devices Market.
The medical device industry is subject to rigorous regulatory standards and approval processes to ensure patient safety and device efficacy. Obtaining regulatory approvals can be time-consuming and costly, slowing down the introduction of new devices to the market.
OPPORTUNITY:
There's potential for developing devices that can be tailored to individual neonatal patients' needs.
Global Expansion & Market Penetration is the opportunity for the Neonatal Respiratory Care Devices Market.
The market has the potential to expand into regions with growing healthcare infrastructure and awareness. Companies can explore opportunities to penetrate new markets and reach a broader range of neonatal care facilities.
CHALLENGES:
Neonates have unique physiological characteristics that make respiratory care more complex.
Ethical & Moral Considerations are the challenges of the Neonatal Respiratory Care Devices Market.
Decisions regarding the intensity of respiratory care for extremely premature or critically ill neonates can be ethically and morally challenging. Balancing the benefits of intervention with potential risks and quality of life considerations is a complex task.
Russia's assault on Ukraine has resulted in the bombing of dozens of hospitals and clinics, as well as regular power outages that can shut off life-saving devices. Medical assistance organisations are employing a Kenyan-made breathing apparatus for preterm babies that works without power to rescue vulnerable newborns in conflict-affected nations. In the field of medical equipment, Russia and Ukraine provide a significant portion of world resources for crude oil, gas as well as some metals. Because US healthcare organisations rely on common raw materials, medical supply prices may grow. Russia generates 18% of the world's natural gas and 13% of its oil. Production disruptions are expected, which might have a direct impact on the global supply chain and plastics pricing. European countries will have to hunt for other suppliers when the supply chains are interrupted as a result of an invasion. As a consequence, natural gas prices are set to rise even further. To offset these costs, producers who purchase raw materials will be allowed to raise their prices. However, healthcare facilities, like their clients, will be affected. During the Russia-Ukraine conflict, the Neonatal Respiratory Care Devices Market increased its product and service prices by 3.4-3.8%.
IMPACT OF ECONOMIC SLOWDOWN
The costs of respiratory support systems are considerable and are sometimes overlooked by clinicians who operate largely at the bedside. A typical ventilator machine costs between $30,100 and $50,000 USD, but a basic continuous positive airway pressure (CPAP) or bi-level positive airway pressure (BiPAP) equipment costs between $1,200 and $12,000. Oxygen blenders and flow metres, as well as certain humidifiers, frequently cost approximately $1,200 USD, which is out of most health facilities' budgets. In 2019, In comparison to high-income and higher-middle-income nations, low-income and low-middle-income countries spent 5.8% of GDP on health, whereas high-income and upper-middle-income countries spent 7.4% of GDP. Given that the average GDP of a high-income country (HIC) is more than 6.1 times that of a low-income nation (LMIC), this disparity is much more pronounced. The financial burden is exacerbated by the need to purchase many devices, as well as the costs of cleaning, maintenance, repairs, and supplies. In the midst of a recession, newborn respiratory care equipment has a lucrative share of up to 2.2-2.8%.
By Product Type
Devices
Accessories & Consumables
By End User
Specialty Clinics
Hospitals
REGIONAL ANALYSIS
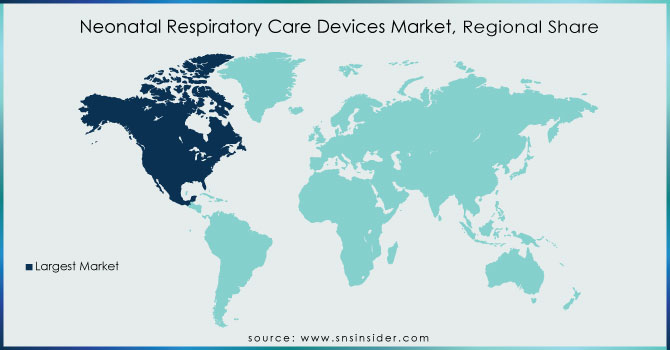
North America: The dominance of North America in the market is owing to the rising frequency of newborn respiratory distress syndrome, along with more NICU hospitalisations due to respiratory problems. Furthermore, the launch of several sophisticated goods by medical device businesses in the United States is a factor contributing to the region's growth.
Asia Pacific: Asia Pacific is expected to have the largest CAGR due to rising demand for accessories and consumables, as well as increased development of healthcare infrastructure to support the establishment of a fully-equipped NICU. This can be attributable to the region's high number of new births as well as a rise in healthcare spending. Furthermore, increased government initiatives in emerging nations such as India are boosting the expansion of the Asia Pacific market.
Get Customized Report as per Your Business Requirement - Request For Customized Report
North America
US
Canada
Mexico
Europe
Eastern Europe
Poland
Romania
Hungary
Turkey
Rest of Eastern Europe
Western Europe
Germany
France
UK
Italy
Spain
Netherlands
Switzerland
Austria
Rest of Western Europe
Asia Pacific
China
India
Japan
South Korea
Vietnam
Singapore
Australia
Rest of Asia Pacific
Middle East & Africa
Middle East
UAE
Egypt
Saudi Arabia
Qatar
Rest of Middle East
Africa
Nigeria
South Africa
Rest of Africa
Latin America
Brazil
Argentina
Colombia
Rest of Latin America
Some major players in Neonatal Respiratory Care Devices Market are Ambu A/S, Cardinal Health, Koninklijke Philips N.V, Drägerwerk AG & Co. KGaA, Medtronic, GE Healthcare, BD, Vyaire, Natus Medical Incorporated, Utah Medical Products, Inc. and other players.
In 2021: SAANS, the world's first portable neonatal CAP device, was introduced by InnAccel for newborns suffering from respiratory distress syndrome. SANS can be employed in a variety of environments and for therapeutic purposes.
In 2020: The FDA approved Drgerwerk AG & CoKGaA's Seattle Positive Airway Pressure System, Plus System for the treatment of infants with pulmonary distress syndrome.
| Report Attributes | Details |
| Market Size in 2023 | US$ 1.82 Bn |
| Market Size by 2032 | US$ 3.10 Bn |
| CAGR | CAGR of 6.1 % From 2024 to 2032 |
| Base Year | 2023 |
| Forecast Period | 2024-2032 |
| Historical Data | 2020-2022 |
| Report Scope & Coverage | Market Size, Segments Analysis, Competitive Landscape, Regional Analysis, DROC & SWOT Analysis, Forecast Outlook |
| Key Segments | • By Product Type (Devices, Accessories & Consumables) • By End User (Specialty Clinics and Hospitals) |
| Regional Analysis/Coverage | North America (US, Canada, Mexico), Europe (Eastern Europe [Poland, Romania, Hungary, Turkey, Rest of Eastern Europe] Western Europe] Germany, France, UK, Italy, Spain, Netherlands, Switzerland, Austria, Rest of Western Europe]). Asia Pacific (China, India, Japan, South Korea, Vietnam, Singapore, Australia, Rest of Asia Pacific), Middle East & Africa (Middle East [UAE, Egypt, Saudi Arabia, Qatar, Rest of Middle East], Africa [Nigeria, South Africa, Rest of Africa], Latin America (Brazil, Argentina, Colombia Rest of Latin America) |
| Company Profiles | Ambu A/S, Cardinal Health, Koninklijke Philips N.V, Drägerwerk AG & Co. KGaA, Medtronic, GE Healthcare, BD, Vyaire, Natus Medical Incorporated, Utah Medical Products, Inc. |
| Key Drivers | • The incidence of premature births has been increasing due to various factors such as maternal age, multiple pregnancies, and medical conditions. • Rising Awareness and Better Healthcare Infrastructure is the driver of the Neonatal Respiratory Care Devices Market. |
| Market Restraints | • Neonatal respiratory care devices can be expensive to develop, manufacture, and purchase. • The stringent Regulatory Approval Process restrains the Neonatal Respiratory Care Devices Market. |
Ans. The Compound Annual Growth rate for Neonatal Respiratory Care Devices Market over the forecast period is 6.1%.
Ans. USD 3.10 billion is the projected Neonatal Respiratory Care Devices Market size of the market by 2032.
Ans. The devices segment is likely to lead this market among product categories over the forecast period.
Ans. Growth in the frequency of newborn jaundice, an increase in the number of preterm deliveries, and the development of novel technology for use in neonatal intensive care units (NICU) are key drivers driving the neonatal critical care equipment market.
Ans. With a market share of roughly 40.25% in 2023, North America led the neonatal critical care equipment market. This is due to strong demand for technologically improved solutions that provide better neonatal healthcare delivery and widespread adoption of the value-based healthcare model.
Table of Contents
1. Introduction
1.1 Market Definition
1.2 Scope
1.3 Research Assumptions
2. Research Methodology
3. Market Dynamics
3.1 Drivers
3.2 Restraints
3.3 Opportunities
3.4 Challenges
4. Impact Analysis
4.1 Impact of Russia-Ukraine War
4.2 Impact of Ongoing Recession
4.2.1 Introduction
4.2.2 Impact on major economies
4.2.2.1 US
4.2.2.2 Canada
4.2.2.3 Germany
4.2.2.4 France
4.2.2.5 United Kingdom
4.2.2.6 China
4.2.2.7 Japan
4.2.2.8 South Korea
4.2.2.9 Rest of the World
5. Value Chain Analysis
6. Porter’s 5 forces model
7. PEST Analysis
8. Neonatal Respiratory Care Devices Market Segmentation, By Product Type
8.1 Devices
8.1.1 Mechanical Ventilators
8.1.2 Extracorporal Membrane Oxygenation (ECMO)
8.1.3 Bubble CAP Systems
8.1.4 Others
8.2 Accessories & Consumables
8.2.1 Breathing Circuits Resuscitators
8.2.2 Nasal Prong Cannula
8.2.3 Masks
8.2.4 Others
9. Neonatal Respiratory Care Devices Market Segmentation, By End User
9.1 Specialty Clinics
9.2 Hospitals
10. Regional Analysis
10.1 Introduction
10.2 North America
10.2.1 North America Neonatal Respiratory Care Devices Market by Country
10.2.2 North America Neonatal Respiratory Care Devices Market by Product Type
10.2.3 North America Neonatal Respiratory Care Devices Market by End User
10.2.4 USA
10.2.4.1 USA Neonatal Respiratory Care Devices Market by Product Type
10.2.4.2 USA Neonatal Respiratory Care Devices Market by End User
10.2.5 Canada
10.2.5.1 Canada Neonatal Respiratory Care Devices Market by Product Type
10.2.5.2 Canada Neonatal Respiratory Care Devices Market by End User
10.2.6 Mexico
10.2.6.1 Mexico Neonatal Respiratory Care Devices Market by Product Type
10.2.6.2 Mexico Neonatal Respiratory Care Devices Market by End User
10.3 Europe
10.3.1 Eastern Europe
10.3.1.1 Eastern Europe Neonatal Respiratory Care Devices Market by Country
10.3.1.2 Eastern Europe Neonatal Respiratory Care Devices Market by Product Type
10.3.1.3 Eastern Europe Neonatal Respiratory Care Devices Market by End User
10.3.1.4 Poland
10.3.1.4.1 Poland Neonatal Respiratory Care Devices Market by Product Type
10.3.1.4.2 Poland Neonatal Respiratory Care Devices Market by End User
10.3.1.5 Romania
10.3.1.5.1 Romania Neonatal Respiratory Care Devices Market by Product Type
10.3.1.5.2 Romania Neonatal Respiratory Care Devices Market by End User
10.3.1.6 Hungary
10.3.1.6.1 Hungary Neonatal Respiratory Care Devices Market by Product Type
10.3.1.6.2 Hungary Neonatal Respiratory Care Devices Market by End User
10.3.1.7 Turkey
10.3.1.7.1 Turkey Neonatal Respiratory Care Devices Market by Product Type
10.3.1.7.2 Turkey Neonatal Respiratory Care Devices Market by End User
10.3.1.8 Rest of Eastern Europe
10.3.1.8.1 Rest of Eastern Europe Neonatal Respiratory Care Devices Market by Product Type
10.3.1.8.2 Rest of Eastern Europe Neonatal Respiratory Care Devices Market by End User
10.3.2 Western Europe
10.3.2.1 Western Europe Neonatal Respiratory Care Devices Market by Country
10.3.2.2 Western Europe Neonatal Respiratory Care Devices Market by Product Type
10.3.2.3 Western Europe Neonatal Respiratory Care Devices Market by End User
10.3.2.4 Germany
10.3.2.4.1 Germany Neonatal Respiratory Care Devices Market by Product Type
10.3.2.4.2 Germany Neonatal Respiratory Care Devices Market by End User
10.3.2.5 France
10.3.2.5.1 France Neonatal Respiratory Care Devices Market by Product Type
10.3.2.5.2 France Neonatal Respiratory Care Devices Market by End User
10.3.2.6 UK
10.3.2.6.1 UK Neonatal Respiratory Care Devices Market by Product Type
10.3.2.6.2 UK Neonatal Respiratory Care Devices Market by End User
10.3.2.7 Italy
10.3.2.7.1 Italy Neonatal Respiratory Care Devices Market by Product Type
10.3.2.7.2 Italy Neonatal Respiratory Care Devices Market by End User
10.3.2.8 Spain
10.3.2.8.1 Spain Neonatal Respiratory Care Devices Market by Product Type
10.3.2.8.2 Spain Neonatal Respiratory Care Devices Market by End User
10.3.2.9 Netherlands
10.3.2.9.1 Netherlands Neonatal Respiratory Care Devices Market by Product Type
10.3.2.9.2 Netherlands Neonatal Respiratory Care Devices Market by End User
10.3.2.10 Switzerland
10.3.2.10.1 Switzerland Neonatal Respiratory Care Devices Market by Product Type
10.3.2.10.2 Switzerland Neonatal Respiratory Care Devices Market by End User
10.3.2.11 Austria
10.3.2.11.1 Austria Neonatal Respiratory Care Devices Market by Product Type
10.3.2.11.2 Austria Neonatal Respiratory Care Devices Market by End User
10.3.2.12 Rest of Western Europe
10.3.2.12.1 Rest of Western Europe Neonatal Respiratory Care Devices Market by Product Type
10.3.2.12.2 Rest of Western Europe Neonatal Respiratory Care Devices Market by End User
10.4 Asia-Pacific
10.4.1 Asia Pacific Neonatal Respiratory Care Devices Market by Country
10.4.2 Asia Pacific Neonatal Respiratory Care Devices Market by Product Type
10.4.3 Asia Pacific Neonatal Respiratory Care Devices Market by End User
10.4.4 China
10.4.4.1 China Neonatal Respiratory Care Devices Market by Product Type
10.4.4.2 China Neonatal Respiratory Care Devices Market by End User
10.4.5 India
10.4.5.1 India Neonatal Respiratory Care Devices Market by Product Type
10.4.5.2 India Neonatal Respiratory Care Devices Market by End User
10.4.6 Japan
10.4.6.1 Japan Neonatal Respiratory Care Devices Market by Product Type
10.4.6.2 Japan Neonatal Respiratory Care Devices Market by End User
10.4.7 South Korea
10.4.7.1 South Korea Neonatal Respiratory Care Devices Market by Product Type
10.4.7.2 South Korea Neonatal Respiratory Care Devices Market by End User
10.4.8 Vietnam
10.4.8.1 Vietnam Neonatal Respiratory Care Devices Market by Product Type
10.4.8.2 Vietnam Neonatal Respiratory Care Devices Market by End User
10.4.9 Singapore
10.4.9.1 Singapore Neonatal Respiratory Care Devices Market by Product Type
10.4.9.2 Singapore Neonatal Respiratory Care Devices Market by End User
10.4.10 Australia
10.4.10.1 Australia Neonatal Respiratory Care Devices Market by Product Type
10.4.10.2 Australia Neonatal Respiratory Care Devices Market by End User
10.4.11 Rest of Asia-Pacific
10.4.11.1 Rest of Asia-Pacific Neonatal Respiratory Care Devices Market by Product Type
10.4.11.2 Rest of Asia-Pacific Neonatal Respiratory Care Devices Market by End User
10.5 Middle East & Africa
10.5.1 Middle East
10.5.1.1 Middle East Neonatal Respiratory Care Devices Market by Country
10.5.1.2 Middle East Neonatal Respiratory Care Devices Market by Product Type
10.5.1.3 Middle East Neonatal Respiratory Care Devices Market by End User
10.5.1.4 UAE
10.5.1.4.1 UAE Neonatal Respiratory Care Devices Market by Product Type
10.5.1.4.2 UAE Neonatal Respiratory Care Devices Market by End User
10.5.1.5 Egypt
10.5.1.5.1 Egypt Neonatal Respiratory Care Devices Market by Product Type
10.5.1.5.2 Egypt Neonatal Respiratory Care Devices Market by End User
10.5.1.6 Saudi Arabia
10.5.1.6.1 Saudi Arabia Neonatal Respiratory Care Devices Market by Product Type
10.5.1.6.2 Saudi Arabia Neonatal Respiratory Care Devices Market by End User
10.5.1.7 Qatar
10.5.1.7.1 Qatar Neonatal Respiratory Care Devices Market by Product Type
10.5.1.7.2 Qatar Neonatal Respiratory Care Devices Market by End User
10.5.1.8 Rest of Middle East
10.5.1.8.1 Rest of Middle East Neonatal Respiratory Care Devices Market by Product Type
10.5.1.8.2 Rest of Middle East Neonatal Respiratory Care Devices Market by End User
10.5.2 Africa
10.5.2.1 Africa Neonatal Respiratory Care Devices Market by Country
10.5.2.2 Africa Neonatal Respiratory Care Devices Market by Product Type
10.5.2.3 Africa Neonatal Respiratory Care Devices Market by End User
10.5.2.4 Nigeria
10.5.2.4.1 Nigeria Neonatal Respiratory Care Devices Market by Product Type
10.5.2.4.2 Nigeria Neonatal Respiratory Care Devices Market by End User
10.5.2.5 South Africa
10.5.2.5.1 South Africa Neonatal Respiratory Care Devices Market by Product Type
10.5.2.5.2 South Africa Neonatal Respiratory Care Devices Market by End User
10.5.2.6 Rest of Africa
10.5.2.6.1 Rest of Africa Neonatal Respiratory Care Devices Market by Product Type
10.5.2.6.2 Rest of Africa Neonatal Respiratory Care Devices Market by End User
10.6 Latin America
10.6.1 Latin America Neonatal Respiratory Care Devices Market by Country
10.6.2 Latin America Neonatal Respiratory Care Devices Market by Product Type
10.6.3 Latin America Neonatal Respiratory Care Devices Market by End User
10.6.4 Brazil
10.6.4.1 Brazil Neonatal Respiratory Care Devices Market by Product Type
10.6.4.2 Brazil Africa Neonatal Respiratory Care Devices Market by End User
10.6.5 Argentina
10.6.5.1 Argentina Neonatal Respiratory Care Devices Market by Product Type
10.6.5.2 Argentina Neonatal Respiratory Care Devices Market by End User
10.6.6 Colombia
10.6.6.1 Colombia Neonatal Respiratory Care Devices Market by Product Type
10.6.6.2 Colombia Neonatal Respiratory Care Devices Market by End User
10.6.7 Rest of Latin America
10.6.7.1 Rest of Latin America Neonatal Respiratory Care Devices Market by Product Type
10.6.7.2 Rest of Latin America Neonatal Respiratory Care Devices Market by End User
11. Company Profile
11.1 Ambu A/S
11.1.1 Company Overview
11.1.2 Financials
11.1.3 Product/Services Offered
11.1.4 SWOT Analysis
11.1.5 The SNS View
11.2 Cardinal Health
11.2.1 Company Overview
11.2.2 Financials
11.2.3 Product/Services Offered
11.2.4 SWOT Analysis
11.2.5 The SNS View
11.3 Koninklijke Philips N.V
11.3.1 Company Overview
11.3.2 Financials
11.3.3 Product/Services Offered
11.3.4 SWOT Analysis
11.3.5 The SNS View
11.4 Drägerwerk AG & Co. KGaA
11.4 Company Overview
11.4.2 Financials
11.4.3 Product/Services Offered
11.4.4 SWOT Analysis
11.4.5 The SNS View
11.5 Medtronic
11.5.1 Company Overview
11.5.2 Financials
11.5.3 Product/Services Offered
11.5.4 SWOT Analysis
11.5.5 The SNS View
11.6 GE Healthcare
11.6.1 Company Overview
11.6.2 Financials
11.6.3 Product/Services Offered
11.6.4 SWOT Analysis
11.6.5 The SNS View
11.7 BD
11.7.1 Company Overview
11.7.2 Financials
11.7.3 Product/Services Offered
11.7.4 SWOT Analysis
11.7.5 The SNS View
11.8 Vyaire
11.8.1 Company Overview
11.8.2 Financials
11.8.3 Product/Services Offered
11.8.4 SWOT Analysis
11.8.5 The SNS View
11.9 Natus Medical Incorporated
11.9.1 Company Overview
11.9.2 Financials
11.9.3 Product/ Services Offered
11.9.4 SWOT Analysis
11.9.5 The SNS View
11.10 Utah Medical Products, Inc.
11.10.1 Company Overview
11.10.2 Financials
11.10.3 Product/Services Offered
11.10.4 SWOT Analysis
11.10.5 The SNS View
12. Competitive Landscape
12.1 Competitive Benchmarking
12.2 Market Share Analysis
12.3 Recent Developments
12.3.1 Industry News
12.3.2 Company News
12.3.3 Mergers & Acquisitions
13. USE Cases and Best Practices
14. Conclusion
An accurate research report requires proper strategizing as well as implementation. There are multiple factors involved in the completion of good and accurate research report and selecting the best methodology to compete the research is the toughest part. Since the research reports we provide play a crucial role in any company’s decision-making process, therefore we at SNS Insider always believe that we should choose the best method which gives us results closer to reality. This allows us to reach at a stage wherein we can provide our clients best and accurate investment to output ratio.
Each report that we prepare takes a timeframe of 350-400 business hours for production. Starting from the selection of titles through a couple of in-depth brain storming session to the final QC process before uploading our titles on our website we dedicate around 350 working hours. The titles are selected based on their current market cap and the foreseen CAGR and growth.
The 5 steps process:
Step 1: Secondary Research:
Secondary Research or Desk Research is as the name suggests is a research process wherein, we collect data through the readily available information. In this process we use various paid and unpaid databases which our team has access to and gather data through the same. This includes examining of listed companies’ annual reports, Journals, SEC filling etc. Apart from this our team has access to various associations across the globe across different industries. Lastly, we have exchange relationships with various university as well as individual libraries.
Step 2: Primary Research
When we talk about primary research, it is a type of study in which the researchers collect relevant data samples directly, rather than relying on previously collected data. This type of research is focused on gaining content specific facts that can be sued to solve specific problems. Since the collected data is fresh and first hand therefore it makes the study more accurate and genuine.
We at SNS Insider have divided Primary Research into 2 parts.
Part 1 wherein we interview the KOLs of major players as well as the upcoming ones across various geographic regions. This allows us to have their view over the market scenario and acts as an important tool to come closer to the accurate market numbers. As many as 45 paid and unpaid primary interviews are taken from both the demand and supply side of the industry to make sure we land at an accurate judgement and analysis of the market.
This step involves the triangulation of data wherein our team analyses the interview transcripts, online survey responses and observation of on filed participants. The below mentioned chart should give a better understanding of the part 1 of the primary interview.
Part 2: In this part of primary research the data collected via secondary research and the part 1 of the primary research is validated with the interviews from individual consultants and subject matter experts.
Consultants are those set of people who have at least 12 years of experience and expertise within the industry whereas Subject Matter Experts are those with at least 15 years of experience behind their back within the same space. The data with the help of two main processes i.e., FGDs (Focused Group Discussions) and IDs (Individual Discussions). This gives us a 3rd party nonbiased primary view of the market scenario making it a more dependable one while collation of the data pointers.
Step 3: Data Bank Validation
Once all the information is collected via primary and secondary sources, we run that information for data validation. At our intelligence centre our research heads track a lot of information related to the market which includes the quarterly reports, the daily stock prices, and other relevant information. Our data bank server gets updated every fortnight and that is how the information which we collected using our primary and secondary information is revalidated in real time.

Step 4: QA/QC Process
After all the data collection and validation our team does a final level of quality check and quality assurance to get rid of any unwanted or undesired mistakes. This might include but not limited to getting rid of the any typos, duplication of numbers or missing of any important information. The people involved in this process include technical content writers, research heads and graphics people. Once this process is completed the title gets uploader on our platform for our clients to read it.
Step 5: Final QC/QA Process:
This is the last process and comes when the client has ordered the study. In this process a final QA/QC is done before the study is emailed to the client. Since we believe in giving our clients a good experience of our research studies, therefore, to make sure that we do not lack at our end in any way humanly possible we do a final round of quality check and then dispatch the study to the client.
The Global eClinical Solutions Market, valued at USD 9.82 Billion in 2023, is projected to grow at a 14.02% CAGR, reaching USD 31.90 Billion by 2032.
Medical Batteries Market was valued at USD 1.96 billion in 2023 and is expected to reach USD 3.33 billion by 2032, growing at a CAGR of 6.03% from 2024-2032.
Disinfection Cap Market Size was valued at USD 368.2 million in 2023 and is expected to reach USD 515.9 million by 2032, growing at a CAGR of 3.84% over the forecast period 2024-2032.
The Newborn Screening Market size was USD 880 million in 2023 and is expected to reach USD 1,710.7 million by 2032, growing at a 7.68% CAGR from 2024 to 2032.
First Aid Market Size was valued at USD 4.66 Billion in 2023 and is expected to reach USD 6.9 billion by 2032, growing at a CAGR of 4.48% over the forecast period 2024-2032.
The Multimodal Imaging Market size was valued at USD 4.43 billion in 2023 and is expected to reach USD 7.43 Billion by 2032, growing at a CAGR of 5.93% during the forecast period of 2024-2032.
Hi! Click one of our member below to chat on Phone