Metastatic Lung Adenocarcinoma Treatment Market Report Scope & Overview:
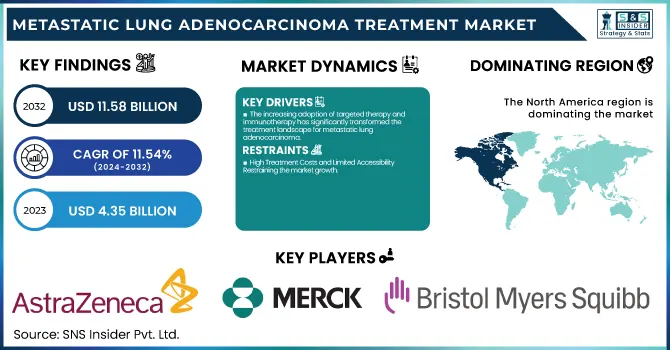
The Metastatic Lung Adenocarcinoma Treatment Market was valued at USD 4.35 billion in 2023 and is expected to reach USD 11.58 billion by 2032, growing at a CAGR of 11.54% from 2024-2032. This report provides information on the Metastatic Lung Adenocarcinoma Treatment Market based on important statistical data. The report presents incidence and prevalence rates for metastatic lung adenocarcinoma, with comprehensive insight into the disease burden. Further, the report reviews prescription trends globally, illustrating treatment choice and adoption rates. Our report also investigates patient volume undergoing treatment, highlighting therapeutic trend shifts. we provide healthcare expenditure distribution by government, commercial, private, and out-of-pocket sources, offering a monetary outlook on the accessibility and affordability of treatment. These findings inform strategic decision-making in the emerging treatment environment.
To Get more information on Metastatic Lung Adenocarcinoma Treatment Market - Request Free Sample Report
Metastatic Lung Adenocarcinoma Treatment Market Dynamics
Drivers
-
The increasing adoption of targeted therapy and immunotherapy has significantly transformed the treatment landscape for metastatic lung adenocarcinoma.
The growing use of targeted therapy and immunotherapy has greatly altered the treatment pattern for metastatic lung adenocarcinoma. Targeted treatments, including EGFR, ALK, and KRAS inhibitors, offer precision therapy by targeting specific cancer-causing mutations. Immunotherapy agents like PD-1/PD-L1 inhibitors, including Bristol-Myers Squibb's Opdivo and Merck's Keytruda, have yielded incredible survival gains. As per a 2023 clinical trial, patients undergoing immunotherapy in combination with chemotherapy had a 30% greater overall survival rate than those undergoing chemotherapy alone. The FDA's February 2024 approval of AstraZeneca's Tagrisso in combination with chemotherapy further underscores the trend toward combination regimens. These developments have driven demand for novel treatment options, enhancing patient outcomes and fueling market growth. The growing number of clinical trials and biomarker-guided therapies keeps reinforcing the implementation of personalized medicine in metastatic lung adenocarcinoma.
-
Rising Incidence of Lung Cancer and Smoking-Related Cases is a key driver for market expansion.
The growing prevalence of lung cancer globally, especially metastatic lung adenocarcinoma, is one of the driving factors for market growth. Lung cancer, as projected by the World Health Organization (WHO), continues to be the most common cause of cancer deaths, with more than 2.2 million new cases in 2023. Nearly 85% of lung cancers are due to continued smoking, which is the main risk factor. Growing exposure to indoor air pollution and workplace carcinogens is also contributing to the prevalence of disease. The American Cancer Society also indicated that non-small cell lung cancer (NSCLC), such as adenocarcinoma, constitutes around 84% of all U.S. cases of lung cancer. Increased awareness and early screening initiatives, coupled with the expanding utilization of next-generation sequencing (NGS) for mutation identification, have improved the earlier treatment and diagnostic planning, which in turn continues to stimulate the use of new therapy options in the market.
Restraint
-
High Treatment Costs and Limited Accessibility Restraining the market growth.
The exorbitant price of metastatic lung adenocarcinoma therapy continues to be a major hindrance to patient access, especially in low- and middle-income countries. Sophisticated treatments like targeted therapies and immunotherapies are not only expensive. For example, AstraZeneca's Tagrisso (osimertinib) costs around USD15,000 a month, while Merck's Keytruda (pembrolizumab) can be over USD150,000 a year. Even in more affluent regions, healthcare reimbursement regulations are different, and thus, treatment is differentially available. An American Society of Clinical Oncology (ASCO) 2023 report reported that close to 20% of American lung cancer patients experience financial constraints in treatment affordability. Moreover, in developing economies, limited biomarker testing availability impinges on the uptake of personalized treatments. All these setbacks, in addition to increasing global healthcare spending, limit patient access to lifesaving drugs, which holds back market growth.
Opportunities
-
The growing advancements in biomarker-driven therapies present a significant opportunity in the metastatic lung adenocarcinoma treatment market.
Personalized medicine is revolutionizing cancer treatment, with targeted treatments such as EGFR, ALK, and KRAS inhibitors demonstrating better patient outcomes. Roche was approved by the FDA for Tecentriq Hybreza, a subcutaneous PD-(L)1 inhibitor, in 2024, significantly shortening administration time. In addition, Merck's Phase 3 trial of MK-1084, a KRAS G12C inhibitor, marks continued innovation in precision oncology. The expanding use of comprehensive genomic profiling and next-generation sequencing (NGS) allows oncologists to personalize therapy successfully. With an increase in clinical trials and partnerships between pharmaceutical companies and diagnostic firms, the incorporation of biomarker-based therapies is likely to improve survival rates and increase treatment options, offering a profitable opportunity for market expansion.
Challenges
-
One of the critical challenges in metastatic lung adenocarcinoma treatment is the emergence of resistance to targeted therapies.
EGFR, ALK, and KRAS inhibitors are highly effective at first, but most patients eventually develop acquired resistance and experience disease progression over time. For instance, research has indicated that close to 60% of EGFR-mutant NSCLC patients develop resistance to frontline osimertinib as a result of secondary mutations or bypass signaling pathways. This requires ongoing research into new-generation inhibitors and combination treatments. Furthermore, tumor heterogeneity complicates the accurate prediction of treatment response. AstraZeneca and Amgen are investing in new therapeutic strategies, but resistance is still challenging to overcome. Without resistance management breakthroughs, patients risk having few long-term treatment options, which is a major challenge for the industry in maintaining the prolonged efficacy of current therapies.
Metastatic Lung Adenocarcinoma Treatment Market Segmentation Analysis
By Treatment
The chemotherapy segment dominated the metastatic lung adenocarcinoma treatment market with a 34.26% market share in 2023 because it has been the gold standard for years, especially among patients who do not have particular genetic mutations. Chemotherapy is still commonly prescribed because it can be used in a wide range of lung cancer subtypes and can effectively decrease tumor burden. It is also frequently combined with targeted therapies or immunotherapies to increase the effectiveness of treatment. The availability and relative affordability of chemotherapy over new medicines have further augmented its ascendancy, particularly where healthcare budgets are low. Irrespective of the breakthrough by targeted medicines and immunotherapeutics, chemotherapy is still the fundamental treatment of choice, even more so among end-stage patients as well as individuals who cannot undergo molecular-targeted medications.
Immunotherapy will be the fastest-growing segment during the forecast years because of its revolutionary role in lung cancer treatment. The launch of immune checkpoint inhibitors such as Merck's Keytruda (pembrolizumab) and Bristol-Myers Squibb's Opdivo (nivolumab) has dramatically enhanced survival rates among patients with metastatic lung adenocarcinoma. Immunotherapy boosts the patient's immune system to recognize and kill cancer cells, with a greater duration of action and fewer adverse effects than conventionally used chemotherapy. As patient selection criteria continue to evolve through research, combination regimens with chemotherapy and targeted agents further drive the acceptance of immunotherapy, positioning it as the most rapidly growing market in the forecast years.
By End Use
The hospitals segment dominated the metastatic lung adenocarcinoma market with around 66.35% market share in 2023 because of their holistic treatment process for cancer patients, from diagnosis to advanced level of treatment options and post-treatment care. Hospitals, mainly large oncology centers and tertiary care facilities, provide a multipronged style of cancer management, bringing under one umbrella chemotherapy, immunotherapy, targeted therapy, and radiotherapy. In addition, hospitals also have access to advanced diagnostic imaging and laboratory equipment to facilitate exact tumor profiling and tailored treatment regimens. Clinics of clinical trials and novel therapies also contribute to patient preference for treatment in hospitals. Additionally, reimbursement policies for healthcare also prefer hospital-based cancer treatment, and hence it becomes the most preferred option for several patients, particularly in developed parts of the world like North America and Europe.
The specialty clinics segment is anticipated to witness the fastest growth in the forecast period as a result of the rising demand for outpatient-based cancer care and personalized medicine. Specialty oncology clinics are more patient-friendly, with greater emphasis on targeted therapies and immunotherapy and fewer hospitalization stays. These clinics provide faster access to oncologists, cutting waiting times for starting treatment. Also, technological advancements in subcutaneous and oral cancer treatments like Roche's Tecentriq Hybreza, enabling quicker delivery outside of hospitals, are making specialist clinics increasingly feasible for the treatment of metastatic lung adenocarcinoma. An escalating number of partnerships between oncology centers and pharmaceutical firms to increase treatment availability, combined with the cost-effectiveness of such clinics, further contributes to the segment's swift growth, especially in developing markets.
Regional Analysis
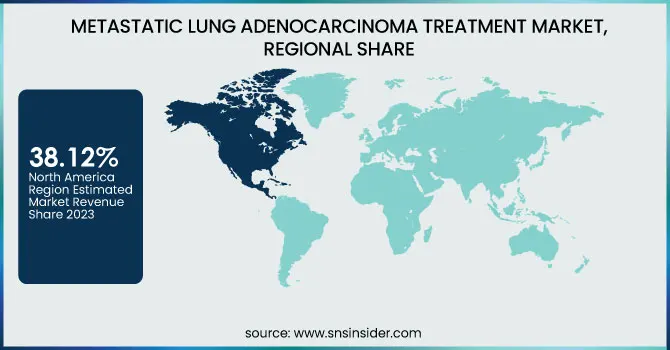
North America dominated the metastatic lung adenocarcinoma treatment market with a 38.12% market share in 2023, owing to its strong established healthcare infrastructure, high adoption rate of innovative therapies, and high investment in cancer research. North America has a high concentration of major pharmaceutical firms like AstraZeneca, Merck, and Pfizer, which are focused on developing and marketing targeted therapies and immunotherapies. The U.S. Food and Drug Administration (FDA) has also been leading in the approval of innovative treatments like AstraZeneca's Tagrisso along with chemotherapy, which was approved in February 2024 under the FLAURA2 trial findings. The greater incidence of lung cancer due to smoking behavior and environmental factors pushes the demand for effective treatments further. Public and private healthcare expenditures also promote patients' access to new treatments, solidifying the leadership of North America in the market.
The Asia Pacific is experiencing the fastest expansion in the metastatic lung adenocarcinoma treatment market with a 12.68% CAGR, because of the growing incidence of lung cancer, augmenting healthcare investment, and improving access to more advanced treatments. China, Japan, and India are increasing their oncology infrastructure to treat cancer and introducing national screening programs to detect lung cancer at an earlier stage. There's also a high growth in clinical trials and regulatory approvals in the region, including China's fast-track nods for new-generation EGFR and ALK inhibitors. In addition, the increasing use of precision medicine and the option for cost-saving biosimilars are fueling market growth. Government efforts to subsidize the treatment of oncology and partnerships between international pharma companies and local biotech firms continue to spur the high development of the market for treating metastatic lung adenocarcinoma in Asia Pacific.
Get Customized Report as per Your Business Requirement - Enquiry Now
Key Players
-
AstraZeneca (Tagrisso, Iressa)
-
Roche (Tarceva, Alecensa)
-
Merck & Co. (Keytruda, Lenvima)
-
Bristol-Myers Squibb (Opdivo, Yervoy)
-
Pfizer (Xalkori, Vizimpro)
-
Novartis (Zykadia, Mekinist)
-
Eli Lilly and Company (Cyramza, Alimta)
-
Johnson & Johnson (Rybrevant, Lazcluze)
-
Amgen (Lumakras, Vectibix)
-
Takeda Pharmaceutical Company (Alunbrig, Mobocertinib)
-
Boehringer Ingelheim (Giotrif, Vargatef)
-
Sanofi (Libtayo, Jevtana)
-
Daiichi Sankyo (Enhertu, Patritumab Deruxtecan)
-
Astellas Pharma (Xospata, Tarceva)
-
GlaxoSmithKline (Zejula, Jemperli)
-
Regeneron Pharmaceuticals (Libtayo, Zaltrap)
-
AbbVie (Rova-T, Imbruvica)
-
Clovis Oncology (Rubraca, Lucitanib)
-
Blueprint Medicines (Gavreto, Ayvakit)
-
BeiGene (Tislelizumab, Pamiparib)
Suppliers (These suppliers play a crucial role in the production, development, and supply chain of metastatic lung adenocarcinoma treatments, including biologics, small-molecule drugs, and targeted therapies.)
-
Lonza Group
-
WuXi AppTec
-
Catalent, Inc.
-
Samsung Biologics
-
Boehringer Ingelheim BioXcellence
-
Patheon (Thermo Fisher Scientific)
-
Samsung Bioepis
-
Fujifilm Diosynth Biotechnologies
-
AGC Biologics
-
Abzena plc
Recent Development
-
February 2024 – AstraZeneca's Tagrisso (osimertinib) with chemotherapy has been approved in the US to treat adult patients with locally advanced or metastatic epidermal growth factor receptor-mutated (EGFRm) non-small cell lung cancer (NSCLC). The approval followed the results of the FLAURA2 clinical trial, which showed that Tagrisso combined with chemotherapy lengthened median progression-free survival by almost nine months versus the standard of care.
-
April 2024 – Merck has started a Phase 3 clinical trial to explore MK-1084, a potential oral KRAS G12C inhibitor, in combination with KEYTRUDA (pembrolizumab) as initial treatment for some patients with metastatic non-small cell lung cancer. The study will evaluate the safety and efficacy of the combination regimen to determine whether it can enhance patient outcomes.
-
September 2024 – Roche reported that the U.S. Food and Drug Administration (FDA) has approved Tecentriq Hybreza (atezolizumab and hyaluronidase-tqjs), the first and only PD-(L)1 inhibitor to be given as a subcutaneous (SC) injection in the U.S. This new form of administration makes it possible to give in about seven minutes, cutting infusion time significantly from the 30-60 minute intravenous (IV) infusion of Tecentriq (atezolizumab). Tecentriq Hybreza will be available for all the prior-approved IV indications in adult patients, such as some lung, liver, skin, and soft tissue cancers.
| Report Attributes | Details |
|---|---|
| Market Size in 2023 | US$ 4.35 billion |
| Market Size by 2032 | US$ 11.58 billion |
| CAGR | CAGR of 11.54% From 2024 to 2032 |
| Base Year | 2023 |
| Forecast Period | 2024-2032 |
| Historical Data | 2020-2022 |
| Report Scope & Coverage | Market Size, Segments Analysis, Competitive Landscape, Regional Analysis, DROC & SWOT Analysis, Forecast Outlook |
| Key Segments | • By Treatment (Chemotherapy, Targeted Therapy, Immunotherapy, Radiation Therapy, Other) • By End Use (Hospitals, Specialty Clinics, Other) |
| Regional Analysis/Coverage | North America (US, Canada, Mexico), Europe (Eastern Europe [Poland, Romania, Hungary, Turkey, Rest of Eastern Europe] Western Europe] Germany, France, UK, Italy, Spain, Netherlands, Switzerland, Austria, Rest of Western Europe]), Asia Pacific (China, India, Japan, South Korea, Vietnam, Singapore, Australia, Rest of Asia Pacific), Middle East & Africa (Middle East [UAE, Egypt, Saudi Arabia, Qatar, Rest of Middle East], Africa [Nigeria, South Africa, Rest of Africa], Latin America (Brazil, Argentina, Colombia, Rest of Latin America) |
| Company Profiles | AstraZeneca, Roche, Merck & Co., Bristol-Myers Squibb, Pfizer, Novartis, Eli Lilly and Company, Johnson & Johnson, Amgen, Takeda Pharmaceutical Company, Boehringer Ingelheim, Sanofi, Daiichi Sankyo, Astellas Pharma, GlaxoSmithKline, Regeneron Pharmaceuticals, AbbVie, Clovis Oncology, Blueprint Medicines, BeiGene, and other players. |

