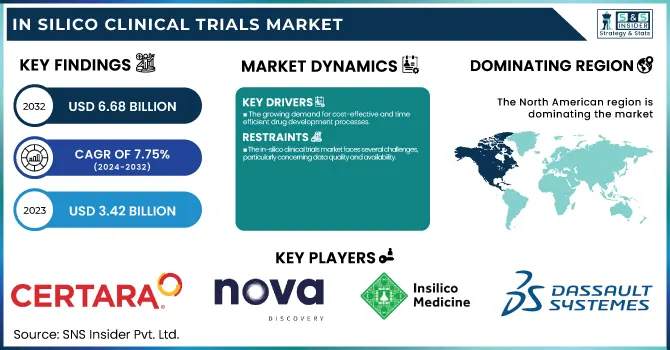
The In Silico Clinical Trials Market was valued at USD 3.42 billion in 2023 and is expected to reach USD 6.68 billion by 2032, growing at a CAGR of 7.75% over the forecast period of 2024-2032. This report analyzes regulatory and approval trends with an emphasis on how changing policies are impacting the use of computational modeling and simulation in clinical trials. The research also discusses technology adoption and integration, emphasizing the increasing use of sophisticated software, AI, and machine learning tools to improve trial accuracy and efficiency. In addition, the report reveals trial simulation insights, highlighting how virtual patient models and predictive analytics are being increasingly used to enhance drug development processes. The report also examines investment trends and funding streams, with venture capital and public-private partnerships fuelling growth in the in-silico sector. Partnerships with biotech startups are at the forefront of developing in silico technologies, driving innovation, and speeding up clinical trial timelines.
To Get more information on In Silico Clinical Trials Market - Request Free Sample Report
The U.S. In Silico Clinical Trials Market was valued at USD 1.11 billion in 2023 and is expected to reach USD 1.98 billion by 2032, growing at a CAGR of 6.66% over the forecast period of 2024-2032. In the United States, the in silico clinical trials market is picking up steam as a result of robust support from the FDA's regulatory favor, increased investments in digital health technologies, and greater emphasis on lowering drug development costs and time-to-market for novel therapies.
Drivers
The growing demand for cost-effective and time-efficient drug development processes.
In silico trials, the quicker simulation of clinical trials and the minimization of the need for large-scale physical trials and extensive patient recruitment. This is especially important against the backdrop of increasing healthcare expenditures and the need for pharmaceutical firms to reduce the drug development timeline. Furthermore, the combination of AI and machine learning technologies adds to the reliability and accuracy of in silico models, and they become all the more desirable. For example, the application of AI-simulation is assisting in the prediction of drug safety and efficacy profiles before clinical trials, thereby decreasing new drug failure rates substantially. Additionally, regulatory bodies such as the FDA are increasingly embracing digital clinical trials, and guidelines are being revised to include in silico approaches. This trend is assisting in increasing the use of such trials. The pandemic caused by COVID-19 also spurred the application of virtual trials, proving the power of in silico approaches for streamlined vaccine development, such as the production of COVID-19 vaccines. These elements all form a supportive environment for the expansion of the market.
Restraints
The in-silico clinical trials market faces several challenges, particularly concerning data quality and availability.
In-silico models depend on high-quality, extensive datasets to effectively simulate real-world conditions, and the absence of standardised, comprehensive clinical data remains a significant barrier. Incomplete or low-quality data can compromise the validity of these models, creating inaccurate trial results and potential regulatory delays. In addition, there is a challenge in getting enough real-world evidence (RWE) to feed these models, as many datasets are dispersed in different healthcare systems and jurisdictions. There is also an absence of universally accepted standards of data integration and validation to makes the problem more complex. Moreover, although in silico trials are becoming increasingly acceptable, most regulatory authorities continue to ask for conventional clinical trial data for licensure, hence restraining the use of digital trials on a large scale. The technology itself also has hindrances in the form of high upfront investment requirements for advanced simulation platforms and a requirement for trained professionals who can effectively use these systems. The sluggish uptake of these technologies in smaller and resource-poor healthcare systems also contributes to the restraint.
Opportunities
The in-silico clinical trials market presents numerous opportunities, particularly in the growing demand for personalized medicine.
With advances in genomic information and precision medicine, the need for clinical trials that can mimic individual patient responses to therapy is growing. In-silico trials are perfect for this, as they can simulate treatment for a given genetic profile, enabling more rapid and personalized drug development. Another huge opportunity is in combining in silico trials with real-world data (RWD). By integrating information from wearable devices, patient monitoring systems, and electronic health records, these trials can provide more realistic simulations with greater accuracy. The capacity to perform hybrid trials, blending in silico models with conventional clinical trials, is also a new possibility. This can streamline the clinical trial process, decreasing time and expense. In addition, emerging markets, where resources for traditional clinical trial infrastructure might be scarce, offer great growth opportunities. Asian-Pacific and Latin American countries, for example, would be well-positioned to use virtual and in silico clinical trials as an alternative to the classical approach since it represents a more effective solution to the worldwide healthcare problem.
Challenges
One of the primary challenges in the in-silico clinical trials market is the regulatory uncertainty surrounding the use of digital simulations in drug approval processes.
Although regulatory authorities such as the FDA and the EMA have progressed in designing guidelines for conducting virtual trials, the absence of universally agreed-on standards and regulations continues to represent an obstacle. Pharmaceutical firms and research institutions consequently may be unwilling to fully exploit the use of in silico trials due to apprehension of potential regulatory problems and drug delays in approval. Moreover, the complexity of in silico models and their integration with current clinical workflows presents a considerable challenge. Many pharmaceutical companies and CROs are still using traditional clinical trial approaches, and the shift to in silico trials involves major technology and training investments. In addition, although in silico trials can limit physical patient recruitment, replicating human variability in simulations is as yet limited. Ensuring that these models reproduce the wide range of patient responses to treatments accurately continues to be a major technical challenge. These problems could inhibit wider acceptance and broader use of in silico clinical trials within the pharmaceutical and medical device sectors.
By Phase
In 2023, the Phase II segment captured the largest market share with a revenue share of 40.6%. The reason for its dominance is mostly because Phase II trials play a vital role in determining whether new treatments work and are safe in a wider population of patients. These trials are instrumental in deciding on the best dosage and further optimizing treatment strategies before transitioning to more expansive testing in Phase III trials. The high percentage of Phase II trials represents the vital role played by Phase II trials in the drug development process, with an increasing focus on expedited drug approvals and precision medicine.
The Phase III segment will be the fastest growing in the market. Phase III trials are the last stage before a drug's introduction into the market, where there is large-scale testing to verify the drug's effectiveness and safety. The demand for effective drugs, coupled with regulatory agencies' efforts toward making more patient access to novel therapies available, are propelling growth in Phase III trials.
By Industry
In 2023, the medical devices segment captured the highest revenue share of 54.6%, indicating its well-established position in clinical trials. The universal adoption of medical devices in most healthcare settings and the fast rate of technological evolution in device innovation have largely been responsible for this leadership. Medical devices are an integral component of contemporary healthcare, and the consistent innovations, especially in diagnostics, wearables, and surgical devices, continue to fuel market growth.
The pharmaceutical sector is expected to record the highest growth in the forecast period. Increased demand for advanced treatments such as biologics, personalized medicine, and gene therapies will fuel pharmaceutical trials' growth. Moreover, pharmaceutical companies' heightened emphasis on the efficiency of drug development and approval processes is translating into an uptrend in clinical trials, thereby fueling the growth of this segment.
By Therapeutic Area
In 2023, the oncology segment represented 22.9% of the overall market revenue, and it was the largest therapeutic area for clinical trials. The widespread incidence of cancer worldwide, combined with the growing need for new and targeted cancer treatments, has played a major role in this segment leading the way. In addition, oncology has been a core focus of investigation for the formulation of immunotherapies, individualized medicines, and targeted medicine, which are likely to continue driving growth in oncology clinical trials.
The infectious disease segment is anticipated to be the most rapidly growing over the forecast period. The COVID-19 pandemic has expedited the demand for quick vaccine and antiviral treatment development, with a focus on infectious disease trials. Due to the growth of emerging diseases and global health threats, the demand for novel therapeutic products in this segment is anticipated to grow, thus driving segment growth.
North America led the in silico clinical trials market in 2023, capturing the highest share of global revenue. North America's leadership is largely due to the strong presence of leading pharmaceutical firms, superior healthcare infrastructure, and high investment in research and development (R&D). The U.S., for instance, has taken a lead in embracing state-of-the-art technologies such as in silico trials because of friendly regulatory authorities such as the FDA, which has come up with guidelines promoting the utilization of computer simulations within clinical trials. Also, the extremely high emphasis on precision medicine, the rising requirement for cost-saving solutions for drug development, and the broad adoption of AI-based tools have further led to the regional market leadership. North America is strengthened by its strong healthcare infrastructure and deeply rooted clinical trial ecosystem, which favors the development of digital trials.
Asia-Pacific is likely to be the most rapidly growing region through the forecast period. This rapid growth can be attributed to the rising number of clinical trials carried out in countries like China, Japan, and India, where there is heavy investment in healthcare innovation. Increased demand for low-cost, effective healthcare solutions, alongside increased adoption of technology and regulatory support for digital health technologies, sees the Asia-Pacific as a leading market driver for growth. The growing healthcare infrastructure and big, heterogeneous patient populations in this part of the world create a great setting for the fast development of in silico trials.

Get Customized Report as per Your Business Requirement - Enquiry Now
Certara, Inc. – Simcyp Simulator, Phoenix WinNonlin
Novadiscovery Sas – NOVA Simulation Platform
Insilico Medicine, Inc. – GENTRL, InSilico AI
Dassault Systèmes SE – BIOVIA, SIMULIA
GNS Healthcare Inc. – REFS (Reverse Engineering and Forward Simulation)
The AnyLogic Company – AnyLogic Simulation Software
InSilicoTrials – InSilicoTrials Platform
Immunetrics Inc. – Immunetrics Modeling Software
Nuventra Pharma Sciences – Pharmacometrics Services
Abzena Ltd. – Abzena Platform
In Oct 2024, Dassault Systèmes unveiled the world’s first guide for the medical device industry on using virtual twins to expedite clinical trials. This guide, developed in collaboration with the U.S. Food and Drug Administration over five years, introduces the ENRICHMENT Playbook, advancing the integration of virtual twins into the regulatory process to enhance patient safety, compliance, and innovation speed.
In Feb 2024, UK-based AdSilico secured USD 4.4 million in funding to advance its virtual trial solution. The platform leverages AI to generate synthetic populations, enabling more time-efficient trials for medical devices. This development aims to streamline the trial process and accelerate product testing.
| Report Attributes | Details |
|---|---|
| Market Size in 2023 | USD 3.42 billion |
| Market Size by 2032 | USD 6.68 billion |
| CAGR | CAGR of 7.75% From 2024 to 2032 |
| Base Year | 2023 |
| Forecast Period | 2024-2032 |
| Historical Data | 2020-2022 |
| Report Scope & Coverage | Market Size, Segments Analysis, Competitive Landscape, Regional Analysis, DROC & SWOT Analysis, Forecast Outlook |
| Key Segments | • By Phase [Phase I, Phase II, Phase III, Phase IV] • By Industry [Medical Devices, Pharmaceutical] • By Therapeutic Area [Oncology, Infectious Disease, Hematology, Cardiology, Dermatology, Neurology, Diabetes, Others] |
| Regional Analysis/Coverage | North America (US, Canada, Mexico), Europe (Eastern Europe [Poland, Romania, Hungary, Turkey, Rest of Eastern Europe] Western Europe] Germany, France, UK, Italy, Spain, Netherlands, Switzerland, Austria, Rest of Western Europe]), Asia Pacific (China, India, Japan, South Korea, Vietnam, Singapore, Australia, Rest of Asia Pacific), Middle East & Africa (Middle East [UAE, Egypt, Saudi Arabia, Qatar, Rest of Middle East], Africa [Nigeria, South Africa, Rest of Africa], Latin America (Brazil, Argentina, Colombia, Rest of Latin America) |
| Company Profiles | Certara, Inc., Novadiscovery Sas, Insilico Medicine, Inc., Dassault Systèmes SE, GNS Healthcare Inc., The AnyLogic Company, InSilicoTrials, Immunetrics Inc., Nuventra Pharma Sciences, Abzena Ltd. |
Ans: The In Silico Clinical Trials Market is projected to grow at a CAGR of 7.75% during the forecast period.
Ans: By 2032, the In Silico Clinical Trials Market is expected to reach USD 6.68 billion, up from USD 3.42 billion in 2023.
Ans: The growing demand for cost-effective and time-efficient drug development processes.
Ans: The in-silico clinical trials market faces several challenges, particularly concerning data quality and availability.
Ans: North America is the dominant region in the In Silico Clinical Trials Market.
1. Introduction
1.1 Market Definition
1.2 Scope (Inclusion and Exclusions)
1.3 Research & Academic Institutes Assumptions
2. Executive Summary
2.1 Market Overview
2.2 Regional Synopsis
2.3 Competitive Summary
3. Research & Academic Institutes Methodology
3.1 Top-Down Approach
3.2 Bottom-up Approach
3.3. Data Validation
3.4 Primary Interviews
4. Market Dynamics Impact Analysis
4.1 Market Driving Factors Analysis
4.1.1 Drivers
4.1.2 Restraints
4.1.3 Opportunities
4.1.4 Challenges
4.2 PESTLE Analysis
4.3 Porter’s Five Forces Model
5. Statistical Insights and Trends Reporting
5.1 Regulatory and Approval Trends
5.2 Technology Adoption and Integration
5.3 Trial Simulation Insights
5.4 Investment Trends and Funding Flow
5.5 Collaboration with Biotech Startups
6. Competitive Landscape
6.1 List of Major Companies, By Region
6.2 Market Share Analysis, By Region
6.3 Product Benchmarking
6.3.1 Product specifications and features
6.3.2 Pricing
6.4 Strategic Initiatives
6.4.1 Marketing and Promotional Activities
6.4.2 Distribution and Supply Chain Strategies
6.4.3 Expansion plans and new Product launches
6.4.4 Strategic partnerships and collaborations
6.5 Technological Advancements
6.6 Market Positioning and Branding
7. In Silico Clinical Trials Market Segmentation, by Phase
7.1 Chapter Overview
7.2 Phase I
7.2.1 Phase I Market Trends Analysis (2020-2032)
7.2.2 Phase I Market Size Estimates and Forecasts to 2032 (USD Billion)
7.3 Phase II
7.3.1 Phase II Market Trends Analysis (2020-2032)
7.3.2 Phase II Market Size Estimates and Forecasts to 2032 (USD Billion)
7.4 Phase III
7.4.1 Phase III Market Trends Analysis (2020-2032)
7.4.2 Phase III Market Size Estimates and Forecasts to 2032 (USD Billion)
7.5 Phase IV
7.5.1 Phase IV Market Trends Analysis (2020-2032)
7.5.2 Phase IV Market Size Estimates and Forecasts to 2032 (USD Billion)
8. In Silico Clinical Trials Market Segmentation, by Industry
8.1 Chapter Overview
8.2 Medical Devices
8.2.1 Medical Devices Market Trends Analysis (2020-2032)
8.2.2 Medical Devices Market Size Estimates and Forecasts to 2032 (USD Billion)
8.3 Pharmaceutical
8.3.1 Pharmaceutical Market Trends Analysis (2020-2032)
8.3.2 Pharmaceutical Market Size Estimates and Forecasts to 2032 (USD Billion)
9. In Silico Clinical Trials Market Segmentation, by Therapeutic Area
9.1 Chapter Overview
9.2 Oncology
9.2.1 Oncology Market Trends Analysis (2020-2032)
9.2.2 Oncology Market Size Estimates and Forecasts to 2032 (USD Billion)
9.3 Infectious Disease
9.3.1 Infectious Disease Market Trends Analysis (2020-2032)
9.3.2 Infectious Disease Market Size Estimates and Forecasts to 2032 (USD Billion)
9.4 Hematology
9.4.1 Hematology Market Trends Analysis (2020-2032)
9.4.2 Hematology Market Size Estimates and Forecasts to 2032 (USD Billion)
9.5 Cardiology
9.5.1 Cardiology Market Trends Analysis (2020-2032)
9.5.2 Cardiology Market Size Estimates and Forecasts to 2032 (USD Billion)
9.6 Dermatology
9.6.1 Dermatology Market Trends Analysis (2020-2032)
9.6.2 Dermatology Market Size Estimates and Forecasts to 2032 (USD Billion)
9.7 Neurology
9.7.1 Neurology Market Trends Analysis (2020-2032)
9.7.2 Neurology Market Size Estimates and Forecasts to 2032 (USD Billion)
9.8 Diabetes
9.8.1 Diabetes Market Trends Analysis (2020-2032)
9.8.2 Diabetes Market Size Estimates and Forecasts to 2032 (USD Billion)
9.9 Others
9.9.1 Others Market Trends Analysis (2020-2032)
9.9.2 Others Market Size Estimates and Forecasts to 2032 (USD Billion)
10. Regional Analysis
10.1 Chapter Overview
10.2 North America
10.2.1 Trends Analysis
10.2.2 North America In Silico Clinical Trials Market Estimates and Forecasts, by Country (2020-2032) (USD Billion)
10.2.3 North America In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.2.4 North America In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.2.5 North America In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.2.6 USA
10.2.6.1 USA In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.2.6.2 USA In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.2.6.3 USA In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.2.7 Canada
10.2.7.1 Canada In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.2.7.2 Canada In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.2.7.3 Canada In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.2.8 Mexico
10.2.8.1 Mexico In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.2.8.2 Mexico In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.2.8.3 Mexico In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.3 Europe
10.3.1 Eastern Europe
10.3.1.1 Trends Analysis
10.3.1.2 Eastern Europe In Silico Clinical Trials Market Estimates and Forecasts, by Country (2020-2032) (USD Billion)
10.3.1.3 Eastern Europe In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.1.4 Eastern Europe In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.3.1.5 Eastern Europe In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.3.1.6 Poland
10.3.1.6.1 Poland In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.1.6.2 Poland In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.3.1.6.3 Poland In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.3.1.7 Romania
10.3.1.7.1 Romania In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.1.7.2 Romania In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.3.1.7.3 Romania In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.3.1.8 Hungary
10.3.1.8.1 Hungary In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.1.8.2 Hungary In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.3.1.8.3 Hungary In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.3.1.9 turkey
10.3.1.9.1 Turkey In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.1.9.2 Turkey In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.3.1.9.3 Turkey In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.3.1.10 Rest of Eastern Europe
10.3.1.10.1 Rest of Eastern Europe In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.1.10.2 Rest of Eastern Europe In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.3.1.10.3 Rest of Eastern Europe In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.3.2 Western Europe
10.3.2.1 Trends Analysis
10.3.2.2 Western Europe In Silico Clinical Trials Market Estimates and Forecasts, by Country (2020-2032) (USD Billion)
10.3.2.3 Western Europe In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.2.4 Western Europe In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.3.2.5 Western Europe In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.3.2.6 Germany
10.3.2.6.1 Germany In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.2.6.2 Germany In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.3.2.6.3 Germany In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.3.2.7 France
10.3.2.7.1 France In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.2.7.2 France In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.3.2.7.3 France In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.3.2.8 UK
10.3.2.8.1 UK In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.2.8.2 UK In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.3.2.8.3 UK In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.3.2.9 Italy
10.3.2.9.1 Italy In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.2.9.2 Italy In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.3.2.9.3 Italy In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.3.2.10 Spain
10.3.2.10.1 Spain In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.2.10.2 Spain In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.3.2.10.3 Spain In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.3.2.11 Netherlands
10.3.2.11.1 Netherlands In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.2.11.2 Netherlands In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.3.2.11.3 Netherlands In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.3.2.12 Switzerland
10.3.2.12.1 Switzerland In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.2.12.2 Switzerland In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.3.2.12.3 Switzerland In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.3.2.13 Austria
10.3.2.13.1 Austria In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.2.13.2 Austria In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.3.2.13.3 Austria In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.3.2.14 Rest of Western Europe
10.3.2.14.1 Rest of Western Europe In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.3.2.14.2 Rest of Western Europe In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.3.2.14.3 Rest of Western Europe In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.4 Asia Pacific
10.4.1 Trends Analysis
10.4.2 Asia Pacific In Silico Clinical Trials Market Estimates and Forecasts, by Country (2020-2032) (USD Billion)
10.4.3 Asia Pacific In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.4.4 Asia Pacific In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.4.5 Asia Pacific In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.4.6 China
10.4.6.1 China In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.4.6.2 China In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.4.6.3 China In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.4.7 India
10.4.7.1 India In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.4.7.2 India In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.4.7.3 India In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.4.8 Japan
10.4.8.1 Japan In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.4.8.2 Japan In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.4.8.3 Japan In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.4.9 South Korea
10.4.9.1 South Korea In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.4.9.2 South Korea In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.4.9.3 South Korea In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.4.10 Vietnam
10.4.10.1 Vietnam In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.4.10.2 Vietnam In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.4.10.3 Vietnam In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.4.11 Singapore
10.4.11.1 Singapore In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.4.11.2 Singapore In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.4.11.3 Singapore In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.4.12 Australia
10.4.12.1 Australia In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.4.12.2 Australia In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.4.12.3 Australia In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.4.13 Rest of Asia Pacific
10.4.13.1 Rest of Asia Pacific In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.4.13.2 Rest of Asia Pacific In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.4.13.3 Rest of Asia Pacific In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.5 Middle East and Africa
10.5.1 Middle East
10.5.1.1 Trends Analysis
10.5.1.2 Middle East In Silico Clinical Trials Market Estimates and Forecasts, by Country (2020-2032) (USD Billion)
10.5.1.3 Middle East In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.5.1.4 Middle East In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.5.1.5 Middle East In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.5.1.6 UAE
10.5.1.6.1 UAE In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.5.1.6.2 UAE In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.5.1.6.3 UAE In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.5.1.7 Egypt
10.5.1.7.1 Egypt In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.5.1.7.2 Egypt In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.5.1.7.3 Egypt In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.5.1.8 Saudi Arabia
10.5.1.8.1 Saudi Arabia In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.5.1.8.2 Saudi Arabia In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.5.1.8.3 Saudi Arabia In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.5.1.9 Qatar
10.5.1.9.1 Qatar In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.5.1.9.2 Qatar In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.5.1.9.3 Qatar In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.5.1.10 Rest of Middle East
10.5.1.10.1 Rest of Middle East In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.5.1.10.2 Rest of Middle East In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.5.1.10.3 Rest of Middle East In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.5.2 Africa
10.5.2.1 Trends Analysis
10.5.2.2 Africa In Silico Clinical Trials Market Estimates and Forecasts, by Country (2020-2032) (USD Billion)
10.5.2.3 Africa In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.5.2.4 Africa In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.5.2.5 Africa In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.5.2.6 South Africa
10.5.2.6.1 South Africa In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.5.2.6.2 South Africa In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.5.2.6.3 South Africa In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.5.2.7 Nigeria
10.5.2.7.1 Nigeria In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.5.2.7.2 Nigeria In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.5.2.7.3 Nigeria In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.5.2.8 Rest of Africa
10.5.2.8.1 Rest of Africa In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.5.2.8.2 Rest of Africa In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.5.2.8.3 Rest of Africa In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.6 Latin America
10.6.1 Trends Analysis
10.6.2 Latin America In Silico Clinical Trials Market Estimates and Forecasts, by Country (2020-2032) (USD Billion)
10.6.3 Latin America In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.6.4 Latin America In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.6.5 Latin America In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.6.6 Brazil
10.6.6.1 Brazil In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.6.6.2 Brazil In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.6.6.3 Brazil In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.6.7 Argentina
10.6.7.1 Argentina In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.6.7.2 Argentina In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.6.7.3 Argentina In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.6.8 Colombia
10.6.8.1 Colombia In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.6.8.2 Colombia In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.6.8.3 Colombia In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
10.6.9 Rest of Latin America
10.6.9.1 Rest of Latin America In Silico Clinical Trials Market Estimates and Forecasts, by Phase (2020-2032) (USD Billion)
10.6.9.2 Rest of Latin America In Silico Clinical Trials Market Estimates and Forecasts, by Industry (2020-2032) (USD Billion)
10.6.9.3 Rest of Latin America In Silico Clinical Trials Market Estimates and Forecasts, by Therapeutic Area (2020-2032) (USD Billion)
11. Company Profiles
11.1 Certara, Inc.
11.1.1 Company Overview
11.1.2 Financial
11.1.3 Product / Services Offered
11.1.4 SWOT Analysis
11.2 Novadiscovery Sas
11.2.1 Company Overview
11.2.2 Financial
11.2.3 Product / Services Offered
11.2.4 SWOT Analysis
11.3 Insilico Medicine, Inc.
11.3.1 Company Overview
11.3.2 Financial
11.3.3 Product / Services Offered
11.3.4 SWOT Analysis
11.4 Dassault Systèmes SE
11.4.1 Company Overview
11.4.2 Financial
11.4.3 Product / Services Offered
11.4.4 SWOT Analysis
11.5 GNS Healthcare Inc.
11.5.1 Company Overview
11.5.2 Financial
11.5.3 Product / Services Offered
11.5.4 SWOT Analysis
11.6 The AnyLogic Company
11.6.1 Company Overview
11.6.2 Financial
11.6.3 Product / Services Offered
11.6.4 SWOT Analysis
11.7 InSilicoTrials
11.7.1 Company Overview
11.7.2 Financial
11.7.3 Product / Services Offered
11.7.4 SWOT Analysis
11.8 Immunetrics Inc.
11.8.1 Company Overview
11.8.2 Financial
11.8.3 Product / Services Offered
11.8.4 SWOT Analysis
11.9 Nuventra Pharma Sciences
11.9.1 Company Overview
11.9.2 Financial
11.9.3 Product / Services Offered
11.9.4 SWOT Analysis
11.10 Abzena Ltd.
11.10.1 Company Overview
11.10.2 Financial
11.10.3 Product / Services Offered
11.10.4 SWOT Analysis
12. Use Cases and Best Practices
13. Conclusion
An accurate research report requires proper strategizing as well as implementation. There are multiple factors involved in the completion of good and accurate research report and selecting the best methodology to compete the research is the toughest part. Since the research reports we provide play a crucial role in any company’s decision-making process, therefore we at SNS Insider always believe that we should choose the best method which gives us results closer to reality. This allows us to reach at a stage wherein we can provide our clients best and accurate investment to output ratio.
Each report that we prepare takes a timeframe of 350-400 business hours for production. Starting from the selection of titles through a couple of in-depth brain storming session to the final QC process before uploading our titles on our website we dedicate around 350 working hours. The titles are selected based on their current market cap and the foreseen CAGR and growth.
The 5 steps process:
Step 1: Secondary Research:
Secondary Research or Desk Research is as the name suggests is a research process wherein, we collect data through the readily available information. In this process we use various paid and unpaid databases which our team has access to and gather data through the same. This includes examining of listed companies’ annual reports, Journals, SEC filling etc. Apart from this our team has access to various associations across the globe across different industries. Lastly, we have exchange relationships with various university as well as individual libraries.
Step 2: Primary Research
When we talk about primary research, it is a type of study in which the researchers collect relevant data samples directly, rather than relying on previously collected data. This type of research is focused on gaining content specific facts that can be sued to solve specific problems. Since the collected data is fresh and first hand therefore it makes the study more accurate and genuine.
We at SNS Insider have divided Primary Research into 2 parts.
Part 1 wherein we interview the KOLs of major players as well as the upcoming ones across various geographic regions. This allows us to have their view over the market scenario and acts as an important tool to come closer to the accurate market numbers. As many as 45 paid and unpaid primary interviews are taken from both the demand and supply side of the industry to make sure we land at an accurate judgement and analysis of the market.
This step involves the triangulation of data wherein our team analyses the interview transcripts, online survey responses and observation of on filed participants. The below mentioned chart should give a better understanding of the part 1 of the primary interview.
Part 2: In this part of primary research the data collected via secondary research and the part 1 of the primary research is validated with the interviews from individual consultants and subject matter experts.
Consultants are those set of people who have at least 12 years of experience and expertise within the industry whereas Subject Matter Experts are those with at least 15 years of experience behind their back within the same space. The data with the help of two main processes i.e., FGDs (Focused Group Discussions) and IDs (Individual Discussions). This gives us a 3rd party nonbiased primary view of the market scenario making it a more dependable one while collation of the data pointers.
Step 3: Data Bank Validation
Once all the information is collected via primary and secondary sources, we run that information for data validation. At our intelligence centre our research heads track a lot of information related to the market which includes the quarterly reports, the daily stock prices, and other relevant information. Our data bank server gets updated every fortnight and that is how the information which we collected using our primary and secondary information is revalidated in real time.

Step 4: QA/QC Process
After all the data collection and validation our team does a final level of quality check and quality assurance to get rid of any unwanted or undesired mistakes. This might include but not limited to getting rid of the any typos, duplication of numbers or missing of any important information. The people involved in this process include technical content writers, research heads and graphics people. Once this process is completed the title gets uploader on our platform for our clients to read it.
Step 5: Final QC/QA Process:
This is the last process and comes when the client has ordered the study. In this process a final QA/QC is done before the study is emailed to the client. Since we believe in giving our clients a good experience of our research studies, therefore, to make sure that we do not lack at our end in any way humanly possible we do a final round of quality check and then dispatch the study to the client.
By Phase
Phase I
Phase II
Phase III
Phase IV
By Industry
Medical Devices
Pharmaceutical
By Therapeutic Area
Oncology
Infectious Disease
Hematology
Cardiology
Dermatology
Neurology
Diabetes
Others
Request for Segment Customization as per your Business Requirement: Segment Customization Request
North America
US
Canada
Mexico
Europe
Eastern Europe
Poland
Romania
Hungary
Turkey
Rest of Eastern Europe
Western Europe
Germany
France
UK
Italy
Spain
Netherlands
Switzerland
Austria
Rest of Western Europe
Asia Pacific
China
India
Japan
South Korea
Vietnam
Singapore
Australia
Rest of Asia Pacific
Middle East & Africa
Middle East
UAE
Egypt
Saudi Arabia
Qatar
Rest of Middle East
Africa
Nigeria
South Africa
Rest of Africa
Latin America
Brazil
Argentina
Colombia
Rest of Latin America
Request for Country Level Research Report: Country Level Customization Request
Available Customization
With the given market data, SNS Insider offers customization as per the company’s specific needs. The following customization options are available for the report:
Detailed Volume Analysis
Criss-Cross segment analysis (e.g. Product X Application)
Competitive Product Benchmarking
Geographic Analysis
Additional countries in any of the regions
Customized Data Representation
Detailed analysis and profiling of additional market players
The Deep Brain Stimulation (DBS) Devices Market Size was valued at USD 1.25 billion in 2023 and is witnessed to reach USD 3.10 billion by 2032 and grow at a CAGR of 11.12% over the forecast period 2024-2032.
The Veterinary Wound Cleansers Market was valued at USD 193 million in 2023 and is expected to reach USD 412.67 million by 2032, growing at a CAGR of 8.8% over the forecast period of 2024-2032.
The Immunoprotein Diagnostic Testing Market size was valued at USD 8.89 billion in 2023, expected to reach USD 17.13 billion by 2032, growing at a 7.54%.
The Orthopedic Joint Replacement Market size was estimated at USD 22.45 billion in 2023 and is expected to reach USD 44.87billion by 2032 at a CAGR of 8% during the forecast period of 2024-2032.
The Dry Age-Related Macular Degeneration Market Size was valued at USD 4.83 billion in 2023 and is expected to reach USD 10.57 billion by 2032 and grow at a CAGR of 9.1% over the forecast period 2024-2032.
The Wellness Supplements Market was valued at USD 245.41 billion in 2023 and is expected to reach USD 470.93 billion by 2032, growing at a CAGR of 7.51% from 2024 to 2032.
Hi! Click one of our member below to chat on Phone