The Breast Cancer Core Needle Biopsy Market was valued at USD 809.40 million in 2023 and is expected to reach USD 1357.70 million by 2032, growing at a CAGR of 8.32% from 2024-2032.
The report presents a detailed review of important statistical information in the Breast Cancer Core Needle Biopsy Market, noting important trends in the industry. It analyzes breast cancer cases involving core needle biopsy incidence and prevalence in 2023, giving a data-informed view of diagnostic demand. The study also assesses the use of image-guided biopsy methods, coupled with regulatory approvals and compliance patterns, to evaluate technological growth and market trends. Additionally, regional healthcare expenditures on breast cancer diagnosis and hospital vs. outpatient use of biopsy equipment provide significant insights into infrastructure and access patterns.
The U.S. Breast Cancer Core Needle Biopsy Market size was USD 264.81 million in 2023 and is expected to reach USD 435.27 million by 2032, growing at a CAGR of 5.69% over the forecast period of 2024-2032.
The U.S. Breast Cancer Core Needle Biopsy Market is experiencing steady growth due to the increasing prevalence of breast cancer and the rising demand for minimally invasive diagnostic procedures. Advancements in imaging technologies, such as ultrasound and MRI-guided biopsies, are enhancing the accuracy and efficiency of core needle biopsies. Additionally, growing awareness about early cancer detection and the adoption of advanced biopsy techniques in healthcare facilities are driving market expansion. The presence of key medical device manufacturers and continuous innovations in biopsy instruments further contribute to the market's growth.
Drivers
Rising Incidence of Breast Cancer Leading to Increased Biopsy Procedures is propelling the market growth.
The increasing incidence of breast cancer is a major growth driver for the breast cancer core needle biopsy market. As per the American Cancer Society (ACS), in 2023, an estimated 297,790 new cases of invasive breast cancer were diagnosed among women in the United States. Early detection is important to enhance survival rates, thus boosting the demand for less invasive diagnostic procedures such as core needle biopsy. The World Health Organization (WHO) underlines that there has been a growth in detections of breast cancer due to programs of screening and heightened awareness, which require accurate and effective biopsy methods. Progress in ultrasound-guided and vacuum-assisted core needle biopsies in the recent past has further improved the accuracy of diagnosis of breast cancer, prompting their extensive use in hospitals and diagnostic centers across the globe.
Ongoing advances in biopsy technology are fueling the use of core needle biopsy in the diagnosis of breast cancer.
The development of vacuum-assisted core needle biopsy (VAB) and image-guided biopsy systems has greatly enhanced accuracy with decreased procedural discomfort. In November 2024, Mammotome introduced the Mammotome AutoCore Single Insertion Core Biopsy System, increasing efficiency through a single-step approach. Besides this, AI-assisted imaging systems are also being coupled with digital breast tomosynthesis (DBT) to better target lesions. All these technologies are simplifying biopsy procedures, eliminating false negatives, and helping diagnose conditions sooner. The surge in demand for minimally invasive and real-time diagnostic methods across cancer screening initiatives is also fuelling the uptake of core needle biopsy procedures and turning them into the go-to alternative to surgery.
Restraint
High Cost and Limited Accessibility in Developing Regions are restraining the market growth.
One of the significant constraints in the Breast Cancer Core Needle Biopsy Market is the expense of biopsy procedures and poor availability in developing countries. Although core needle biopsy (CNB) is less invasive and an accurate diagnostic procedure, the expense of image-guided biopsies, advanced vacuum-assisted biopsy systems, and related pathology tests can be too high for patients in low- and middle-income countries (LMICs). As per the WHO, more than 70% of deaths from breast cancer take place in these areas due to delayed diagnosis and economic conditions. Furthermore, the absence of specialized diagnostic facilities, trained personnel, and an insufficient healthcare infrastructure also limits widespread use of core needle biopsy. The above limitations necessitate the use of low-cost biopsy technologies and government-sponsored screening programs for the improvement of early detection of breast cancer in underprivileged groups.
Opportunities
The use of artificial intelligence (AI) and digital pathology is a big opportunity in the Breast Cancer Core Needle Biopsy Market.
AI-based diagnostic equipment can improve the accuracy of biopsy interpretation, computerize image analysis, and help detect malignancies at an early stage. AI-assisted pathology enhanced breast cancer detection rates by 12-15% compared to conventional methods, as per a 2023 JAMA study. Moreover, digital pathology platforms facilitate distant diagnostics, where skilled pathologists can examine biopsy samples remotely, enhancing access to high-quality cancer diagnostics in underserved areas. With the growing usage of AI-based biopsy interpretation software and machine learning algorithms, market participants are anticipated to leverage AI-based innovations to streamline workflow, minimize diagnostic mistakes, and maximize treatment planning for breast cancer patients.
Challenges
Variability in Biopsy Accuracy and False-Negative Rates is challenging the market growth.
A primary problem with the Breast Cancer Core Needle Biopsy Market is inconsistency in biopsy precision as well as false-negative rates. Although core needle biopsy (CNB) is common in breast lesion diagnosis, research suggests that false-negative rates vary between 2% and 10%, contingent on tumor heterogeneity, technical procedure, and operator proficiency. In situations where small or non-palpable lesions are not well sampled, CNB can result in inconclusive results, delayed diagnoses, and the possibility of disease progression. Another drawback is that image-guided biopsies like ultrasound- or MRI-guided CNB also need specialized training, and variability in the execution of the procedure can compromise diagnostic reliability. Resolving this challenge mandates technical innovations in biopsy needle design, artificial intelligence-based biopsy navigation, and uniform training programs for pathologists and radiologists to enhance diagnostic accuracy.
By Technology
The Ultrasound-Based Breast Biopsy segment dominated the breast cancer core needle biopsy market with a 40.12% market share in 2023 because of its high availability, affordability, and real-time imaging. Ultrasound-guided procedures are more accessible, less painful for patients, and quicker compared to other technologies such as stereotactic and MRI-guided biopsies, and hence are the most preferred by both patients and healthcare professionals. Furthermore, ultrasound does not use ionizing radiation, as opposed to mammography-based biopsies, which is especially useful for younger women and patients who need repeated imaging. The increasing demand for minimally invasive therapies, rising incidence of breast cancer, and technological improvement in high-resolution ultrasound imaging further drove the dominance of the segment. The American College of Radiology (ACR) and the National Comprehensive Cancer Network (NCCN) endorse ultrasound-guided biopsies as the first-line strategy for breast lesions suspected to have cancer, further enhancing their universal acceptance in clinical practice.
By End-Use
The Hospitals & Diagnostic Laboratories segment dominated the Breast Cancer Core Needle Biopsy Market with 58.31% market share in 2023 because of the large patient influx, presence of specialized oncologists and radiologists, and sophisticated diagnostic facilities. These centers are the main referral points for breast cancer screening, biopsy procedures, and follow-up diagnostics, ensuring early and accurate detection of the disease. Moreover, the existence of integrated healthcare systems, government-financed screening programs, and insurance reimbursement for diagnostic procedures led more patients to opt for biopsies in hospitals and accredited diagnostic laboratories. The rising prevalence of breast cancer globally and increasing knowledge about early detection further extended the segment's dominance in the market.
The Academic & Research Institutes segment will see the fastest growth during the forecast period, owing to increased investment in cancer research, biopsy technology advancements, and clinical trials on early and non-invasive breast cancer detection. Growing partnerships between universities, biotech companies, and medical device manufacturers are propelling innovation in precision biopsy methods, AI-based imaging, and liquid biopsy options. Additionally, government and private financing programs for breast cancer screening innovation, biomarker research, and personalized medicine are driving the research applications of core needle biopsy devices at an accelerated rate. The need for enhancing diagnostic precision and less invasive biopsy techniques will persistently drive this segment's growth, and hence, it is a major driver of future market growth.
North America dominated the breast cancer core needle biopsy market with a 45.24% market share in 2024 based on its superior health infrastructure, heavy breast cancer patient burden, and first-time movers in newer biopsy devices. As stated by the American Cancer Society, the United States alone reported nearly 297,790 newly diagnosed breast cancers in 2023, placing demands on precision diagnostic modalities like the core needle biopsy (CNB) devices. Also, the availability of major medical device companies, strong reimbursement policies, and high prevalence of image-guided biopsy procedures further consolidate the region's leadership. Government support in the form of the National Cancer Institute's (NCI) funding for breast cancer research and early detection programs has also helped in the increasing use of CNB devices. Additionally, enhanced patient awareness, availability to specialty diagnostic centers, and ongoing technological innovation make North America the market leader.
The Asia Pacific region is witnessing the fastest growth in the breast cancer core needle biopsy market with 5.31% CAGR throughout the forecast period because of the growing prevalence of breast cancer, rising healthcare spending, and improving access to diagnostic care. These nations, such as China, India, and Japan, are seeing an increase in breast cancer cases, primarily due to changes in lifestyle, delayed childbearing, and demographic aging. The World Health Organization (WHO) reports that breast cancer is the leading female cancer in Asia and demands a greater use of early diagnostic aids like CNB. Also, governments are investing in cancer screening programs, and private healthcare sectors are increasing diagnostic imaging centers, fueling market growth. The growing use of minimally invasive biopsy procedures, increased medical tourism, and increased awareness of breast cancer detection drive the market in the region.
Hologic, Inc. (Eviva Breast Biopsy System, Celero Vacuum-Assisted Breast Biopsy Device)
Becton, Dickinson and Company (BD) (EnCor Ultra Breast Biopsy System, EnCor Enspire Biopsy System)
Mammotome (Mammotome Biopsy System, Mammotome Revolve Dual Vacuum-Assisted Biopsy System)
Resitu Medical (Resitu Breast Biopsy Device, Core Needle Biopsy Device)
Siemens Healthineers (MAMMOMAT Inspiration Biopsy Unit, MAMMOMAT Revelation Biopsy System)
Stryker Corporation (Core Needle Biopsy Devices, Vacuum-Assisted Biopsy Systems)
Cook Medical (EchoTip Ultra Breast Biopsy Needle, Quick-Core Biopsy Needle)
Argon Medical Devices, Inc. (SuperCore Biopsy Instrument, Tru-Core II Automatic Biopsy Instrument)
B. Braun Melsungen AG (Sterican Biopsy Needles, Vacuette Multiple Use Drawing Needle)
Cardinal Health (Achieve Automatic Biopsy Device, Temno Biopsy System)
Medtronic plc (Osteo-Site Bone Biopsy Needle, Kyphon Biopsy Device)
CareFusion Corporation (Max-Core Disposable Core Biopsy Instrument, Mission Disposable Core Biopsy Instrument)
SOMATEX Medical Technologies GmbH (SOMATEX Vacuum-Assisted Breast Biopsy System, SOMATEX Core Needle Biopsy Device)
Devicor Medical Products, Inc. (Neoprobe Gamma Detection System, Mammotome Elite Biopsy System)
Fujifilm Holdings Corporation (Fujifilm Biopsy System, Aspire Breast Biopsy Unit)
GE Healthcare (Senographe Pristina Biopsy, SenoClaire Biopsy System)
Philips Healthcare (MicroDose SI Biopsy, Affiniti 70 Biopsy Guidance)
Nikon Corporation (Nikon Biopsy Imaging Systems, Eclipse Ci-L Plus Biopsy Microscope)
Hitachi Medical Systems (HI VISION Preirus Biopsy, ARIETTA 850 Biopsy System)
Planmed Oy (Planmed Nuance Biopsy Unit, Planmed Clarity Guide)
Suppliers (These suppliers provide key raw materials such as medical-grade plastics, polymers, and coatings used in the manufacturing of biopsy needles, vacuum-assisted devices, and imaging components) in the Breast Cancer Core Needle Biopsy Market
Mitsubishi Chemical Holdings Corporation
3M Company
Covestro AG
DuPont de Nemours, Inc.
Evonik Industries AG
Toray Industries, Inc.
BASF SE
Celanese Corporation
Eastman Chemical Company
Sumitomo Chemical Co., Ltd.
November 2024 – Mammotome unveiled the introduction of the Mammotome AutoCore Single Insertion Core Biopsy System, the first automated spring-loaded core needle biopsy system to be launched in the marketplace. This new product launch signifies Mammotome's continued dedication to furthering breast biopsy technology and optimizing patient care results.
In December 2024, Hologic presented new research at the RSNA's 110th Scientific Assembly and Annual Meeting. The study emphasized that Hologic Genius AI Detection 2.0 software achieves consistent performance across a wide range of patient populations, reaffirming the company's commitment to reducing bias in breast cancer detection.
| Report Attributes | Details |
|---|---|
| Market Size in 2023 | US$ 809.40 million |
| Market Size by 2032 | US$ 1357.70 million |
| CAGR | CAGR of 5.93% From 2024 to 2032 |
| Base Year | 2023 |
| Forecast Period | 2024-2032 |
| Historical Data | 2020-2022 |
| Report Scope & Coverage | Market Size, Segments Analysis, Competitive Landscape, Regional Analysis, DROC & SWOT Analysis, Forecast Outlook |
| Key Segments | • By Technology (MRI-Based Breast Biopsy, Ultrasound-Based Breast Biopsy, Mammography-Based (Stereotactic) Breast Biopsy, CT-Based Breast Biopsy, Other Image-Based Breast Biopsy) • By End-use (Hospitals & Diagnostic Laboratories, Pharmaceutical & Biotechnology Companies, Academic & Research Institutes) |
| Regional Analysis/Coverage | North America (US, Canada, Mexico), Europe (Eastern Europe [Poland, Romania, Hungary, Turkey, Rest of Eastern Europe] Western Europe] Germany, France, UK, Italy, Spain, Netherlands, Switzerland, Austria, Rest of Western Europe]), Asia Pacific (China, India, Japan, South Korea, Vietnam, Singapore, Australia, Rest of Asia Pacific), Middle East & Africa (Middle East [UAE, Egypt, Saudi Arabia, Qatar, Rest of Middle East], Africa [Nigeria, South Africa, Rest of Africa], Latin America (Brazil, Argentina, Colombia, Rest of Latin America) |
| Company Profiles | Hologic, Becton, Dickinson and Company (BD), Mammotome, Resitu Medical, Siemens Healthineers, Stryker Corporation, Cook Medical, Argon Medical Devices, B. Braun Melsungen AG, Cardinal Health, Medtronic, CareFusion Corporation, SOMATEX Medical Technologies GmbH, Devicor Medical Products, Fujifilm Holdings Corporation, GE Healthcare, Philips Healthcare, Nikon Corporation, Hitachi Medical Systems, Planmed Oy, and other players. |
Ans: The Breast Cancer Core Needle Biopsy Market is expected to grow at a CAGR of 5.93% from 2024-2032.
Ans: The Breast Cancer Core Needle Biopsy Market was USD 809.40 million in 2023 and is expected to reach USD 1357.70 million by 2032.
Ans: Ongoing advances in biopsy technology are fueling the use of core needle biopsy in the diagnosis of breast cancer.
Ans: The “Ultrasound-Based Breast Biopsy” segment dominated the Breast Cancer Core Needle Biopsy Market.
Ans: North America dominated the Breast Cancer Core Needle Biopsy Market in 2023.
Table of Contents:
1. Introduction
1.1 Market Definition
1.2 Scope (Inclusion and Exclusions)
1.3 Research Assumptions
2. Executive Summary
2.1 Market Overview
2.2 Regional Synopsis
2.3 Competitive Summary
3. Research Methodology
3.1 Top-Down Approach
3.2 Bottom-up Approach
3.3. Data Validation
3.4 Primary Interviews
4. Market Dynamics Impact Analysis
4.1 Market Driving Factors Analysis
4.1.2 Drivers
4.1.2 Restraints
4.1.3 Opportunities
4.1.4 Challenges
4.2 PESTLE Analysis
5. Statistical Insights and Trends Reporting
5.1 Incidence and Prevalence of Breast Cancer Requiring Core Needle Biopsy (2023)
5.2 Core Needle Biopsy Device Volume, by Region (2020-2032)
5.3 Adoption of Image-Guided Core Needle Biopsy Techniques (2023-2032)
5.4 Regulatory Approvals and Compliance Trends (2023-2032)
5.5 Healthcare Spending on Breast Cancer Diagnostics, by Region (2023)
6. Competitive Landscape
6.1 List of Major Companies, By Region
6.2 Market Share Analysis, By Region
6.3 Product Benchmarking
6.3.1 Product specifications and features
6.3.2 Pricing
6.4 Strategic Initiatives
6.4.1 Marketing and promotional activities
6.4.2 Distribution and Supply Chain Strategies
6.4.3 Expansion plans and new product launches
6.4.4 Strategic partnerships and collaborations
6.5 Technological Advancements
6.6 Market Positioning and Branding
7. Breast Cancer Core Needle Biopsy Market Segmentation, By Technology
7.1 Chapter Overview
7.2 MRI-Based Breast Biopsy
7.2.1 MRI-Based Breast Biopsy Market Trends Analysis (2020-2032)
7.2.2 MRI-Based Breast Biopsy Market Size Estimates and Forecasts to 2032 (USD Million)
7.3 Ultrasound-Based Breast Biopsy
7.3.1 Ultrasound-Based Breast Biopsy Market Trends Analysis (2020-2032)
7.3.2 Ultrasound-Based Breast Biopsy Market Size Estimates and Forecasts to 2032 (USD Million)
7.4 Mammography-Based (Stereotactic) Breast Biopsy
7.4.1 Mammography-Based (Stereotactic) Breast Biopsy Market Trends Analysis (2020-2032)
7.4.2 Mammography-Based (Stereotactic) Breast Biopsy Market Size Estimates and Forecasts to 2032 (USD Million)
7.5 CT-Based Breast Biopsy
7.5.1 CT-Based Breast Biopsy Market Trends Analysis (2020-2032)
7.5.2 CT-Based Breast Biopsy Market Size Estimates and Forecasts to 2032 (USD Million)
7.6 Other Image-Based Breast Biopsy
7.6.1 Other Image-Based Breast Biopsy Market Trends Analysis (2020-2032)
7.6.2 Other Image-Based Breast Biopsy Market Size Estimates and Forecasts to 2032 (USD Million)
8. Breast Cancer Core Needle Biopsy Market Segmentation, By End User
8.1 Chapter Overview
8.2 Hospitals & Diagnostic Laboratories
8.2.1 Hospitals & Diagnostic Laboratories Market Trends Analysis (2020-2032)
8.2.2 Hospitals & Diagnostic Laboratories Market Size Estimates and Forecasts to 2032 (USD Million)
8.3 Pharmaceutical & Biotechnology companies
8.3.1 Pharmaceutical & Biotechnology Companies Market Trends Analysis (2020-2032)
8.3.2 Pharmaceutical & Biotechnology companies Market Size Estimates and Forecasts to 2032 (USD Million)
8.4 Academic & Research Institutes
8.4.1 Academic & Research Institutes Market Trends Analysis (2020-2032)
8.4.2 Academic & Research Institutes Market Size Estimates and Forecasts to 2032 (USD Million)
9. Regional Analysis
9.1 Chapter Overview
9.2 North America
9.2.1 Trends Analysis
9.2.2 North America Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Country (2020-2032) (USD Million)
9.2.3 North America Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.2.4 North America Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.2.5 USA
9.2.5.1 USA Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.2.5.2 USA Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.2.6 Canada
9.2.6.1 Canada Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.2.6.2 Canada Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.2.7 Mexico
9.2.7.1 Mexico Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.2.7.2 Mexico Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.3 Europe
9.3.1 Eastern Europe
9.3.1.1 Trends Analysis
9.3.1.2 Eastern Europe Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Country (2020-2032) (USD Million)
9.3.1.3 Eastern Europe Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.3.1.4 Eastern Europe Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.3.1.5 Poland
9.3.1.5.1 Poland Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.3.1.5.2 Poland Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.3.1.6 Romania
9.3.1.6.1 Romania Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.3.1.6.2 Romania Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.3.1.7 Hungary
9.3.1.7.1 Hungary Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.3.1.7.2 Hungary Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.3.1.8 Turkey
9.3.1.8.1 Turkey Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.3.1.8.2 Turkey Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.3.1.9 Rest of Eastern Europe
9.3.1.9.1 Rest of Eastern Europe Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.3.1.9.2 Rest of Eastern Europe Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.3.2 Western Europe
9.3.2.1 Trends Analysis
9.3.2.2 Western Europe Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Country (2020-2032) (USD Million)
9.3.2.3 Western Europe Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.3.2.4 Western Europe Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.3.2.5 Germany
9.3.2.5.1 Germany Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.3.2.5.2 Germany Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.3.2.6 France
9.3.2.6.1 France Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.3.2.6.2 France Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.3.2.7 UK
9.3.2.7.1 UK Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.3.2.7.2 UK Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.3.2.8 Italy
9.3.2.8.1 Italy Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.3.2.8.2 Italy Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.3.2.9 Spain
9.3.2.9.1 Spain Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.3.2.9.2 Spain Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.3.2.10 Netherlands
9.3.2.10.1 Netherlands Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.3.2.10.2 Netherlands Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.3.2.11 Switzerland
9.3.2.11.1 Switzerland Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.3.2.11.2 Switzerland Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.3.2.12 Austria
9.3.2.12.1 Austria Healthcare Predictive Analytic Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.3.2.12.2 Austria Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.3.2.13 Rest of Western Europe
9.3.2.13.1 Rest of Western Europe Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.3.2.13.2 Rest of Western Europe Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.4 Asia Pacific
9.4.1 Trends Analysis
9.4.2 Asia Pacific Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Country (2020-2032) (USD Million)
9.4.3 Asia Pacific Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.4.4 Asia Pacific Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.4.5 China
9.4.5.1 China Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.4.5.2 China Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.4.6 India
9.4.5.1 India Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.4.5.2 India Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.4.5 Japan
9.4.5.1 Japan Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.4.5.2 Japan Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.4.6 South Korea
9.4.6.1 South Korea Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.4.6.2 South Korea Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.4.7 Vietnam
9.4.7.1 Vietnam Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.2.7.2 Vietnam Healthcare Predictive Analytic Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.4.8 Singapore
9.4.8.1 Singapore Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.4.8.2 Singapore Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.4.9 Australia
9.4.9.1 Australia Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.4.9.2 Australia Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.4.10 Rest of Asia Pacific
9.4.10.1 Rest of Asia Pacific Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.4.10.2 Rest of Asia Pacific Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.5 Middle East and Africa
9.5.1 Middle East
9.5.1.1 Trends Analysis
9.5.1.2 Middle East Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Country (2020-2032) (USD Million)
9.5.1.3 Middle East Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.5.1.4 Middle East Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.5.1.5 UAE
9.5.1.5.1 UAE Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.5.1.5.2 UAE Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.5.1.6 Egypt
9.5.1.6.1 Egypt Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.5.1.6.2 Egypt Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.5.1.7 Saudi Arabia
9.5.1.7.1 Saudi Arabia Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.5.1.7.2 Saudi Arabia Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.5.1.8 Qatar
9.5.1.8.1 Qatar Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.5.1.8.2 Qatar Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.5.1.9 Rest of Middle East
9.5.1.9.1 Rest of Middle East Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.5.1.9.2 Rest of Middle East Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.5.2 Africa
9.5.2.1 Trends Analysis
9.5.2.2 Africa Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Country (2020-2032) (USD Million)
9.5.2.3 Africa Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.5.2.4 Africa Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.5.2.5 South Africa
9.5.2.5.1 South Africa Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.5.2.5.2 South Africa Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.5.2.6 Nigeria
9.5.2.6.1 Nigeria Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.5.2.6.2 Nigeria Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.5.2.7 Rest of Africa
9.5.2.7.1 Rest of Africa Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.5.2.7.2 Rest of Africa Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.6 Latin America
9.6.1 Trends Analysis
9.6.2 Latin America Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Country (2020-2032) (USD Million)
9.6.3 Latin America Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.6.4 Latin America Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.6.5 Brazil
9.6.5.1 Brazil Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.6.5.2 Brazil Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.6.6 Argentina
9.6.6.1 Argentina Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.6.6.2 Argentina Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.6.7 Colombia
9.6.7.1 Colombia Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.6.7.2 Colombia Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
9.6.8 Rest of Latin America
9.6.8.1 Rest of Latin America Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by Technology (2020-2032) (USD Million)
9.6.8.2 Rest of Latin America Breast Cancer Core Needle Biopsy Market Estimates and Forecasts, by End-Use (2020-2032) (USD Million)
10. Company Profiles
10.1 Hologic, Inc.
10.1.1 Company Overview
10.1.2 Financial
10.1.3 Products/ Services Offered
110.1.4 SWOT Analysis
10.2 Becton, Dickinson and Company
10.2.1 Company Overview
10.2.2 Financial
10.2.3 Products/ Services Offered
10.2.4 SWOT Analysis
10.3 Mammotome
10.3.1 Company Overview
10.3.2 Financial
10.3.3 Products/ Services Offered
10.3.4 SWOT Analysis
10.4 Resitu Medical
10.4.1 Company Overview
10.4.2 Financial
10.4.3 Products/ Services Offered
10.4.4 SWOT Analysis
10.5 Siemens Healthineers
10.5.1 Company Overview
10.5.2 Financial
10.5.3 Products/ Services Offered
10.5.4 SWOT Analysis
10.6 Stryker Corporation
10.6.1 Company Overview
10.6.2 Financial
10.6.3 Products/ Services Offered
10.6.4 SWOT Analysis
10.7 Cook Medical
10.7.1 Company Overview
10.7.2 Financial
10.7.3 Products/ Services Offered
10.7.4 SWOT Analysis
10.8 Argon Medical Devices, Inc.
10.8.1 Company Overview
10.8.2 Financial
10.8.3 Products/ Services Offered
10.8.4 SWOT Analysis
10.9 B. Braun Melsungen AG
10.9.1 Company Overview
10.9.2 Financial
10.9.3 Products/ Services Offered
10.9.4 SWOT Analysis
10.10 Cardinal Health
10.9.1 Company Overview
10.9.2 Financial
10.9.3 Products/ Services Offered
10.9.4 SWOT Analysis
11. Use Cases and Best Practices
12. Conclusion
An accurate research report requires proper strategizing as well as implementation. There are multiple factors involved in the completion of good and accurate research report and selecting the best methodology to compete the research is the toughest part. Since the research reports we provide play a crucial role in any company’s decision-making process, therefore we at SNS Insider always believe that we should choose the best method which gives us results closer to reality. This allows us to reach at a stage wherein we can provide our clients best and accurate investment to output ratio.
Each report that we prepare takes a timeframe of 350-400 business hours for production. Starting from the selection of titles through a couple of in-depth brain storming session to the final QC process before uploading our titles on our website we dedicate around 350 working hours. The titles are selected based on their current market cap and the foreseen CAGR and growth.
The 5 steps process:
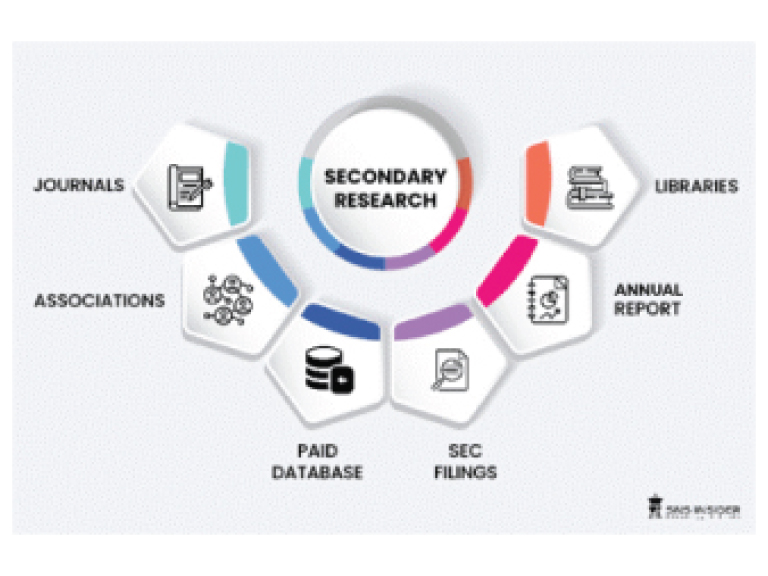
Step 1: Secondary Research:
Secondary Research or Desk Research is as the name suggests is a research process wherein, we collect data through the readily available information. In this process we use various paid and unpaid databases which our team has access to and gather data through the same. This includes examining of listed companies’ annual reports, Journals, SEC filling etc. Apart from this our team has access to various associations across the globe across different industries. Lastly, we have exchange relationships with various university as well as individual libraries.
Step 2: Primary Research
When we talk about primary research, it is a type of study in which the researchers collect relevant data samples directly, rather than relying on previously collected data. This type of research is focused on gaining content specific facts that can be sued to solve specific problems. Since the collected data is fresh and first hand therefore it makes the study more accurate and genuine.
We at SNS Insider have divided Primary Research into 2 parts.
Part 1 wherein we interview the KOLs of major players as well as the upcoming ones across various geographic regions. This allows us to have their view over the market scenario and acts as an important tool to come closer to the accurate market numbers. As many as 45 paid and unpaid primary interviews are taken from both the demand and supply side of the industry to make sure we land at an accurate judgement and analysis of the market.
This step involves the triangulation of data wherein our team analyses the interview transcripts, online survey responses and observation of on filed participants. The below mentioned chart should give a better understanding of the part 1 of the primary interview.
Part 2: In this part of primary research the data collected via secondary research and the part 1 of the primary research is validated with the interviews from individual consultants and subject matter experts.
Consultants are those set of people who have at least 12 years of experience and expertise within the industry whereas Subject Matter Experts are those with at least 15 years of experience behind their back within the same space. The data with the help of two main processes i.e., FGDs (Focused Group Discussions) and IDs (Individual Discussions). This gives us a 3rd party nonbiased primary view of the market scenario making it a more dependable one while collation of the data pointers.
Step 3: Data Bank Validation
Once all the information is collected via primary and secondary sources, we run that information for data validation. At our intelligence centre our research heads track a lot of information related to the market which includes the quarterly reports, the daily stock prices, and other relevant information. Our data bank server gets updated every fortnight and that is how the information which we collected using our primary and secondary information is revalidated in real time.

Step 4: QA/QC Process
After all the data collection and validation our team does a final level of quality check and quality assurance to get rid of any unwanted or undesired mistakes. This might include but not limited to getting rid of the any typos, duplication of numbers or missing of any important information. The people involved in this process include technical content writers, research heads and graphics people. Once this process is completed the title gets uploader on our platform for our clients to read it.
Step 5: Final QC/QA Process:
This is the last process and comes when the client has ordered the study. In this process a final QA/QC is done before the study is emailed to the client. Since we believe in giving our clients a good experience of our research studies, therefore, to make sure that we do not lack at our end in any way humanly possible we do a final round of quality check and then dispatch the study to the client.
Breast Cancer Core Needle Biopsy Market Key Segments:
By Technology
MRI-Based Breast Biopsy
Ultrasound-Based Breast Biopsy
Mammography-Based (Stereotactic) Breast Biopsy
CT-Based Breast Biopsy
Other Image-Based Breast Biopsy
By End-Use
Hospitals & Diagnostic Laboratories
Pharmaceutical & Biotechnology companies
Academic & Research Institutes
Request for Segment Customization as per your Business Requirement: Segment Customization Request
Regional Coverage:
North America
US
Canada
Mexico
Europe
Eastern Europe
Poland
Romania
Hungary
Turkey
Rest of Eastern Europe
Western Europe
Germany
France
UK
Italy
Spain
Netherlands
Switzerland
Austria
Rest of Western Europe
Asia Pacific
China
India
Japan
South Korea
Vietnam
Singapore
Australia
Rest of Asia Pacific
Middle East & Africa
Middle East
UAE
Egypt
Saudi Arabia
Qatar
Rest of Middle East
Africa
Nigeria
South Africa
Rest of Africa
Latin America
Brazil
Argentina
Colombia
Rest of Latin America
Request for Country Level Research Report: Country Level Customization Request
Available Customization
With the given market data, SNS Insider offers customization as per the company’s specific needs. The following customization options are available for the report:
Detailed Volume Analysis
Criss-Cross segment analysis (e.g. Product X Application)
Competitive Product Benchmarking
Geographic Analysis
Additional countries in any of the regions
Customized Data Representation
Detailed analysis and profiling of additional market players
The Injection Pen Market was valued at USD 40.6 billion in 2023 and is projected to reach USD 73.8 billion by 2032, with a growing CAGR of 6.8% from 2024 to 2032.
The Healthcare Education Market size was valued at USD 110 billion in 2023 and is expected to reach USD 280.6 billion by 2032 and grow at a CAGR of 11.0% over the forecast period 2024-2032.
The Serum-Free Media Market Size was valued at USD 1.7 billion in 2023 and is expected to grow at a CAGR of 13.0% to reach USD 5.1 billion by 2032.
The carrier screening market valued at USD 2.26 Billion in 2023, projected to reach USD 11.44 Billion by 2032, growing at a CAGR of 19.76% from 2024–2032.
The global antibiotics market size was USD 47.23 Billion in 2023 & is expected to reach USD 65.23 billion by 2032 at a CAGR of 3.70%.
The Cardiac Implants Market size was valued at USD 38.1 billion in 2023 and is expected to reach USD 79.2 billion by 2032, growing at a CAGR of 8.5% during the forecast period from 2024 to 2032.
Hi! Click one of our member below to chat on Phone