The Blood-Based Biomarker for Sports Medicine Market was valued at USD 727.18 million in 2023 and is expected to reach USD 1347.68 million by 2032, growing at a CAGR of 7.11% over the forecast period of 2024-2032. This report indicates blood-based biomarker testing trends by geography, driven by growing awareness of preventing injury, optimizing performance, and early diagnosis of musculoskeletal disorders. The research analyzes healthcare expenditure on biomarker testing, with increasing investment from sports federations, rehabilitation clinics, and high-performance training centers. It also analyzes adoption trends by sport type, with endurance sports, contact sports, and strength sports exhibiting different levels of demand for biomarker monitoring. The study explores technical progress in areas such as AI-powered analytics and newer biomarker panels, improving diagnosis accuracy and recovery interventions. Regulatory and compliance trends significantly contribute to market dynamics, including the standardization of test protocols and the consideration of ethical implications of sports performance tracking. Additionally, R&D efforts and clinical trials continue to inform innovation, offering evidence-based information on biomarker use in injury risk assessment and recovery tracking.
The U.S. Blood-Based Biomarker for Sports Medicine Market was valued at USD 214.29 million in 2023 and is expected to reach USD 359.07 million by 2032, growing at a CAGR of 5.92% over the forecast period of 2024-2032. In the US, the market is growing with more funding in sports science, more partnerships of biotech companies with sporting organizations, and a high emphasis on customized performance analytics to enhance athlete health and longevity.
Drivers
The rising adoption of blood-based biomarkers in sports medicine is driven by their increasing role in injury prevention, recovery monitoring, and performance optimization.
The increasing incidence of sports injuries, with more than 8.6 million sports injuries reported each year in the U.S. alone, has spurred demand for early and accurate diagnostic methods. Biomarkers such as CK, CRP, and IL-6 offer real-time information on muscle damage, inflammation, and metabolic stress, enabling athletes and coaches to adjust training programs accordingly. Advances in biomarker technology, point-of-care diagnostics, and AI-driven diagnostic solutions are also improving accessibility and efficiency. In addition, increased knowledge among professional and amateur sports players regarding the advantages of biomarker-based evaluation is increasing market penetration. International sport governing bodies such as FIFA and the NBA are more actively implementing biomarker tests into regular monitoring programs of the health of their players. The demand is further driven by convergence with wearable health technologies and blood biomarker tests to enable round-the-clock tracking of athletes' performance and recovery factors. Consequently, sports doctors and rehabilitation specialists are increasingly turning to biomarker-based diagnostics to maximize injury management protocols, minimize recovery time, and maximize overall performance. These drivers continue to drive the market forward, making biomarker-based sports diagnostics a key component of contemporary athletic medicine.
Restraints
The blood-based biomarker market for sports medicine faces significant challenges, primarily due to high costs and limited insurance coverage.
Most biomarker tests are costly, with advanced panels ranging between USD 200 to USD 500 per test, which may limit their usage across the broad population, especially among recreational athletes and small sport organizations. The lack of standardization in reference values for biomarkers is a significant hindrance, as this could vary by age, gender, and physical fitness, thereby creating inconsistent meanings. Regulatory barriers and complicated approval procedures also delay market growth, as novel biomarker-based tests need extensive clinical validation and FDA approvals prior to commercialization. In addition, limited awareness among non-elite athletes and overall sports medicine professionals limits the scope of the market. Although elite sports teams and research institutions are investing heavily in biomarker-based diagnostics, uptake is comparatively low in amateur and grassroots sports environments, owing to financial and logistical limitations. Concerns regarding data privacy linked to biomarker testing also restrict adoption, as individuals are not likely to provide intimate health information. These restrictions in total impede the mass adoption of blood-based biomarkers in sports medicine, necessitating stakeholders in the industry to address cost reduction, regulatory convergence, and wider education schemes.
Opportunities
The blood-based biomarker market for sports medicine presents numerous opportunities, especially with the growing integration of AI, machine learning, and big data analytics in sports diagnostics.
Emerging fast, point-of-care biomarker tests facilitate in real time, and monitoring has become much easier for the assessment of recovery, fatigue, and injury risks for athletes at any moment. Improvements in microfluidic devices and non-invasive biomarker detection would make the market newsworthy through the minimization of venous blood draws and make biomarker testing convenient and easy. Furthermore, rising partnerships among biotechnology firms, sporting organizations, and research institutions are fueling novel biomarker findings with athlete-specific applications. The increased popularity of personalized medicine for sports also offers a potential for biomarker-based performance enhancement programs. Firms are introducing home-testing biomarker kits so that athletes can keep track of their well-being without recurrent hospital visits. In addition, regulatory authorities of professional sports leagues are looking into making biomarker-based testing compulsory for injury prevention programs, yet another growth driver for the market. The growing application of biomarkers in anti-doping campaigns to ensure the safety of athletes and prevent any kind of unfair benefit further supports demand. With ongoing innovation, increasing regulatory backing, and greater availability, the market is ready for tremendous growth across professional and amateur sports segments.
Challenges
The blood-based biomarker market in sports medicine faces several challenges, particularly regarding test accuracy, ethical concerns, and regulatory complexities.
One of the primary issues is that there are no universally accepted thresholds for biomarkers, and therefore, inconsistent diagnoses may result, influencing clinical decision-making. Biomarker levels can also vary because of factors independent of sports injury, including diet, hydration status, and genetic makeup, creating doubts about test reliability. Ethical concerns also exist, as sporting organizations and teams can abuse biomarker information to make career-changing decisions for players, resulting in issues of privacy and consent. Additionally, the market is hampered by regulatory differences between regions, as biomarker tests have to comply with different approval processes, complicating global commercialization. Slow uptake among general practitioners and non-specialist healthcare professionals is another significant challenge, as they might not be trained to interpret biomarkers. While top sports teams spend vast amounts on biomarker-based performance evaluation, smaller groups of organizations and lone athletes usually can't afford, or gain access to, economical and trustworthy testing solutions.
By Type
In 2023, the Creatine Kinase (CK) segment led the blood-based biomarker for sports medicine market with a 15.7% share due to its pivotal role in diagnosing muscle damage, overtraining, and exertional rhabdomyolysis. CK testing is extensively applied in elite sports, rehabilitation clinics, and performance monitoring laboratories to evaluate muscle stress and recovery, thus being the most established biomarker in the market. Its widespread adoption by sports physicians and trainers has made it the dominant segment.
The C-Reactive Protein (CRP) segment was the most rapidly growing as a result of its increased application in the detection of inflammation, prevention of injury, and management of recovery. CRP is becoming more popular for its capability to identify early stages of muscle inflammation and overuse injuries, which has resulted in its extensive use in sports medicine clinics and high-performance training programs. The demand is also driven by the increasing focus on preventing injury and ensuring athlete well-being.
The blood-based biomarker for sports medicine market is witnessing strong growth across major regions, with North America leading the market due to the high adoption of advanced sports diagnostics, the presence of elite sports organizations, and well-established healthcare infrastructure. The United States accounts for the largest share, driven by widespread biomarker testing among professional leagues like the NFL, NBA, and MLB, along with growing investments in athlete health monitoring and sports rehabilitation programs. Europe follows closely, with countries like Germany, the UK, and France witnessing increased adoption due to the rising emphasis on player safety and injury prevention in football (soccer) leagues such as the English Premier League and Bundesliga.
Asia-Pacific is the fastest-growing region, fueled by the expansion of sports science programs, rising participation in professional sports, and increasing government initiatives to enhance athlete performance. Countries like China, Japan, and India are investing heavily in sports medicine research and biomarker-based diagnostics, with China alone seeing a 27% increase in sports-related healthcare spending over the past five years. The rising popularity of endurance sports, combat sports, and esports in Asia is further driving demand for biomarker-driven performance optimization. Latin America and the Middle East & Africa are also showing steady growth, with increasing sports infrastructure and healthcare advancements supporting market expansion.
Abbott – Architect C System, Total Bilirubin2 assay
BIOMÉRIEUX – VIDAS TBI test
F. Hoffmann-La Roche Ltd. – Elecsys β-Amyloid (1-42) CSF II, Elecsys Phospho-Tau (181P) CSF, Benzodiazepines II test
ARUP Laboratories – Comprehensive diagnostic testing services
Siemens Healthineers AG – CardioPhase hsCRP test, ADVIA Centaur immunoassay system, Atellica Solution, Aptio Automation
RayBiotech, Inc. – ELISA kits, protein arrays
Thermo Fisher Scientific, Inc. – DRI Tricyclics Serum Tox Assay, Microgenics products
Bio-Rad Laboratories, Inc. – Bio-Plex multiplex immunoassay system, QX200 Droplet Digital PCR System
Beckman Coulter, Inc. – Access Total βhCG (5th IS), Access Vitamin B12 assay
Randox Laboratories Ltd. – Comprehensive diagnostic solutions
In Dec 2024, Fujirebio Holdings, Inc. and Eisai Co., Ltd. signed a memorandum of understanding for joint research and social implementation of novel blood-based biomarkers targeting neurodegenerative diseases. This collaboration aims to advance biomarker-based diagnostics for early disease detection and management.
In Oct 2024, Sunbird Bio presented new clinical data showcasing its proprietary alpha-synuclein (α-synuclein) blood-based biomarker technology, which accurately detects Parkinson’s disease through a simple blood draw. The findings, shared at the CTAD conference on October 31, highlight the potential for blood-based diagnosis of multiple neurodegenerative diseases with high accuracy.
| Report Attributes | Details |
|---|---|
| Market Size in 2023 | USD 727.18 million |
| Market Size by 2032 | USD 1347.68 million |
| CAGR | CAGR of 7.11% From 2024 to 2032 |
| Base Year | 2023 |
| Forecast Period | 2024-2032 |
| Historical Data | 2020-2022 |
| Report Scope & Coverage | Market Size, Segments Analysis, Competitive Landscape, Regional Analysis, DROC & SWOT Analysis, Forecast Outlook |
| Key Segments | • By Type [CK, Myoglobin, Lactate, WBC, Urea, CRP, Lipid and Protein Hydroperoxides, IL-6, Others] |
| Regional Analysis/Coverage | North America (US, Canada, Mexico), Europe (Eastern Europe [Poland, Romania, Hungary, Turkey, Rest of Eastern Europe] Western Europe] Germany, France, UK, Italy, Spain, Netherlands, Switzerland, Austria, Rest of Western Europe]), Asia Pacific (China, India, Japan, South Korea, Vietnam, Singapore, Australia, Rest of Asia Pacific), Middle East & Africa (Middle East [UAE, Egypt, Saudi Arabia, Qatar, Rest of Middle East], Africa [Nigeria, South Africa, Rest of Africa], Latin America (Brazil, Argentina, Colombia, Rest of Latin America) |
| Company Profiles | Abbott, BIOMÉRIEUX, F. Hoffmann-La Roche Ltd., ARUP Laboratories, Siemens Healthineers AG, RayBiotech, Inc., Thermo Fisher Scientific, Inc., Bio-Rad Laboratories, Inc., Beckman Coulter, Inc., Randox Laboratories Ltd. |
Ans: The Blood-Based Biomarker for Sports Medicine market is anticipated to grow at a CAGR of 7.11% from 2024 to 2032.
Ans: The market is expected to reach USD 1347.68 million by 2032, increasing from USD 727.18 million in 2023.
Ans: The rising adoption of blood-based biomarkers in sports medicine is driven by their increasing role in injury prevention, recovery monitoring, and performance optimization.
Ans: The blood-based biomarker market for sports medicine faces significant challenges, primarily due to high costs and limited insurance coverage.
Ans: North America dominated the Blood-Based Biomarker for Sports Medicine market.
Table of Contents
1. Introduction
1.1 Market Definition
1.2 Scope (Inclusion and Exclusions)
1.3 Research Assumptions
2. Executive Summary
2.1 Market Overview
2.2 Regional Synopsis
2.3 Competitive Summary
3. Research Methodology
3.1 Top-Down Approach
3.2 Bottom-up Approach
3.3. Data Validation
3.4 Primary Interviews
4. Market Dynamics Impact Analysis
4.1 Market Driving Factors Analysis
4.1.1 Drivers
4.1.2 Restraints
4.1.3 Opportunities
4.1.4 Challenges
4.2 PESTLE Analysis
4.3 Porter’s Five Forces Model
5. Statistical Insights and Trends Reporting
5.1 Blood-Based Biomarker Testing Trends (2023), by Region
5.2 Healthcare Spending on Blood-Based Biomarker Testing (2023)
5.3 Adoption Trends by Sports Category (2023-2032)
5.4 Technological Advancements in Blood-Based Biomarker Testing (2020-2032)
5.5 Regulatory and Compliance Trends (2023-2032)
5.6 Research and Clinical Trials Insights (2023-2032)
6. Competitive Landscape
6.1 List of Major Companies, By Region
6.2 Market Share Analysis, By Region
6.3 Product Benchmarking
6.3.1 Product specifications and features
6.3.2 Pricing
6.4 Strategic Initiatives
6.4.1 Marketing and promotional activities
6.4.2 Distribution and Supply Chain Strategies
6.4.3 Expansion plans and new product launches
6.4.4 Strategic partnerships and collaborations
6.5 Technological Advancements
6.6 Market Positioning and Branding
7. Blood-Based Biomarker for Sports Medicine Market Segmentation, By Type
7.1 Chapter Overview
7.2 CK
7.2.1 CK Market Trends Analysis (2020-2032)
7.2.2 CK Market Size Estimates and Forecasts to 2032 (USD Million)
7.3 Myoglobin
7.3.1 Myoglobin Market Trends Analysis (2020-2032)
7.3.2 Myoglobin Market Size Estimates and Forecasts to 2032 (USD Million)
7.4 Lactate
7.4.1 Lactate Market Trends Analysis (2020-2032)
7.4.2 Lactate Market Size Estimates and Forecasts to 2032 (USD Million)
7.5 WBC
7.5.1 WBC Market Trends Analysis (2020-2032)
7.5.2 WBC Market Size Estimates and Forecasts to 2032 (USD Million)
7.6 Urea
7.6.1 Urea Market Trends Analysis (2020-2032)
7.6.2 Urea Market Size Estimates and Forecasts to 2032 (USD Million)
7.7 CRP
7.7.1 CRP Market Trends Analysis (2020-2032)
7.7.2 CRP Market Size Estimates and Forecasts to 2032 (USD Million)
7.8 Lipid and Protein Hydroperoxides
7.8.1 Lipid and Protein Hydroperoxides Market Trends Analysis (2020-2032)
7.8.2 Lipid and Protein Hydroperoxides Market Size Estimates and Forecasts to 2032 (USD Million)
7.9 IL-6
7.9.1 IL-6 Market Trends Analysis (2020-2032)
7.9.2 IL-6 Market Size Estimates and Forecasts to 2032 (USD Million)
7.10 Others
7.10.1 Others Market Trends Analysis (2020-2032)
7.10.2 Others Market Size Estimates and Forecasts to 2032 (USD Million)
8. Regional Analysis
8.1 Chapter Overview
8.2 North America
8.2.1 Trends Analysis
8.2.2 North America Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, by Country (2020-2032) (USD Million)
8.2.3 North America Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.2.2 USA
8.2.2.1 USA Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.2.3 Canada
8.2.3.1 Canada Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.2.4 Mexico
8.2.4.1 Mexico Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.3 Europe
8.3.1 Eastern Europe
8.3.1.1 Trends Analysis
8.3.1.2 Eastern Europe Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, by Country (2020-2032) (USD Million)
8.3.1.3 Eastern Europe Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.3.1.4 Poland
8.3.1.4.1 Poland Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.3.1.5 Romania
8.3.1.5.1 Romania Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.3.1.6 Hungary
10.3.1.8.1 Hungary Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.3.1.7 Turkey
8.3.1.7.1 Turkey Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.3.1.8 Rest of Eastern Europe
8.3.1.8.1 Rest of Eastern Europe Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.3.2 Western Europe
8.3.2.1 Trends Analysis
8.3.2.2 Western Europe Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, by Country (2020-2032) (USD Million)
8.3.2.3 Western Europe Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.3.2.4 Germany
8.3.2.4.1 Germany Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.3.2.5 France
8.3.2.5.1 France Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.3.2.6 UK
8.3.2.6.1 UK Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.3.2.7 Italy
8.3.2.7.1 Italy Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.3.2.8 Spain
8.3.2.8.1 Spain Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.3.2.9 Netherlands
8.3.2.9.1 Netherlands Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.3.2.10 Switzerland
8.3.2.10.1 Switzerland Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.3.2.11 Austria
8.3.2.11.1 Austria Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.3.2.12 Rest of Western Europe
8.3.2.12.1 Rest of Western Europe Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.4 Asia Pacific
8.4.1 Trends Analysis
8.4.2 Asia Pacific Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, by Country (2020-2032) (USD Million)
8.4.3 Asia Pacific Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.4.4 China
8.4.4.1 China Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.4.5 India
8.4.5.1 India Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.4.6 Japan
8.4.6.1 Japan Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.4.7 South Korea
8.4.7.1 South Korea Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.4.8 Vietnam
8.4.8.1 Vietnam Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.4.9 Singapore
8.4.9.1 Singapore Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.4.10 Australia
8.4.10.1 Australia Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.4.11 Rest of Asia Pacific
8.4.11.1 Rest of Asia Pacific Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.5 Middle East and Africa
8.5.1 Middle East
8.5.1.1 Trends Analysis
8.5.1.2 Middle East Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, by Country (2020-2032) (USD Million)
10.5.1.3 Middle East Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.5.1.4 UAE
8.5.1.4.1 UAE Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.5.1.5 Egypt
8.5.1.5.1 Egypt Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.5.1.6 Saudi Arabia
8.5.1.6.1 Saudi Arabia Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.5.1.7 Qatar
8.5.1.7.1 Qatar Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.5.1.8 Rest of Middle East
8.5.1.8.1 Rest of Middle East Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.5.2 Africa
8.5.2.1 Trends Analysis
8.5.2.2 Africa Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, by Country (2020-2032) (USD Million)
8.5.2.3 Africa Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.5.2.4 South Africa
8.5.2.4.1 South Africa Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.5.2.5 Nigeria
8.5.2.5.1 Nigeria Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.5.2.6 Rest of Africa
8.5.2.6.1 Rest of Africa Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.6 Latin America
8.6.1 Trends Analysis
8.6.2 Latin America Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, by Country (2020-2032) (USD Million)
8.6.3 Latin America Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.6.4 Brazil
8.6.4.1 Brazil Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.6.5 Argentina
8.6.5.1 Argentina Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.6.6 Colombia
8.6.6.1 Colombia Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
8.6.7 Rest of Latin America
8.6.7.1 Rest of Latin America Blood-Based Biomarker for Sports Medicine Market Estimates and Forecasts, By Type (2020-2032) (USD Million)
9. Company Profiles
9.1 Abbott
9.1.1 Company Overview
9.1.2 Financial
9.1.3 Products/ Services Offered
9.1.4 SWOT Analysis
9.2 BIOMÉRIEUX
9.2.1 Company Overview
9.2.2 Financial
9.2.3 Products/ Services Offered
9.2.4 SWOT Analysis
9.3 F. Hoffmann-La Roche Ltd.
9.3.1 Company Overview
9.3.2 Financial
9.3.3 Products/ Services Offered
9.3.4 SWOT Analysis
9.4 ARUP Laboratories
9.4.1 Company Overview
9.4.2 Financial
9.4.3 Products/ Services Offered
9.4.4 SWOT Analysis
9.5 Siemens Healthineers AG
9.5.1 Company Overview
9.5.2 Financial
9.5.3 Products/ Services Offered
9.5.4 SWOT Analysis
9.6 RayBiotech, Inc.
9.6.1 Company Overview
9.6.2 Financial
9.6.3 Products/ Services Offered
9.6.4 SWOT Analysis
9.7 Thermo Fisher Scientific, Inc.
9.7.1 Company Overview
9.7.2 Financial
9.7.3 Products/ Services Offered
9.7.4 SWOT Analysis
9.8 Bio-Rad Laboratories, Inc.
9.8.1 Company Overview
9.8.2 Financial
9.8.3 Products/ Services Offered
9.8.4 SWOT Analysis
9.9 Beckman Coulter, Inc.
9.9.1 Company Overview
9.9.2 Financial
9.9.3 Products/ Services Offered
9.9.4 SWOT Analysis
9.10 Randox Laboratories Ltd.
9.10.1 Company Overview
9.10.2 Financial
9.10.3 Products/ Services Offered
9.10.4 SWOT Analysis
10. Use Cases and Best Practices
11. Conclusion
An accurate research report requires proper strategizing as well as implementation. There are multiple factors involved in the completion of good and accurate research report and selecting the best methodology to compete the research is the toughest part. Since the research reports we provide play a crucial role in any company’s decision-making process, therefore we at SNS Insider always believe that we should choose the best method which gives us results closer to reality. This allows us to reach at a stage wherein we can provide our clients best and accurate investment to output ratio.
Each report that we prepare takes a timeframe of 350-400 business hours for production. Starting from the selection of titles through a couple of in-depth brain storming session to the final QC process before uploading our titles on our website we dedicate around 350 working hours. The titles are selected based on their current market cap and the foreseen CAGR and growth.
The 5 steps process:
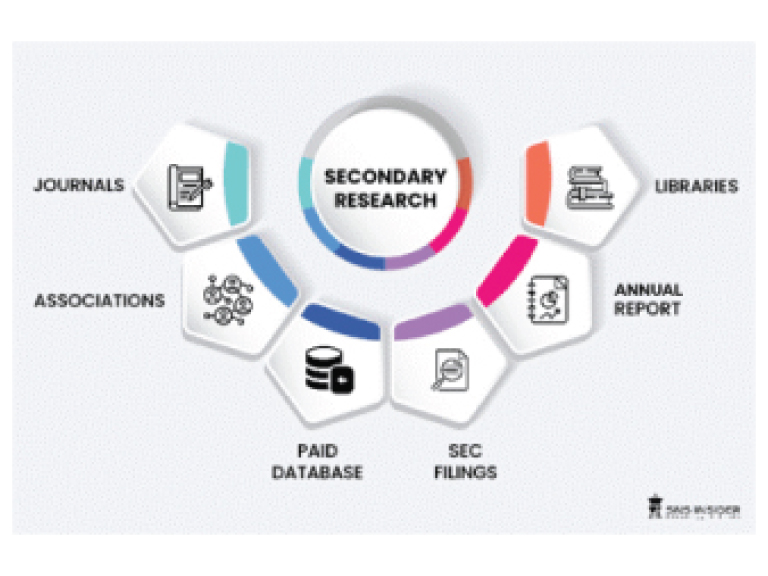
Step 1: Secondary Research:
Secondary Research or Desk Research is as the name suggests is a research process wherein, we collect data through the readily available information. In this process we use various paid and unpaid databases which our team has access to and gather data through the same. This includes examining of listed companies’ annual reports, Journals, SEC filling etc. Apart from this our team has access to various associations across the globe across different industries. Lastly, we have exchange relationships with various university as well as individual libraries.
Step 2: Primary Research
When we talk about primary research, it is a type of study in which the researchers collect relevant data samples directly, rather than relying on previously collected data. This type of research is focused on gaining content specific facts that can be sued to solve specific problems. Since the collected data is fresh and first hand therefore it makes the study more accurate and genuine.
We at SNS Insider have divided Primary Research into 2 parts.
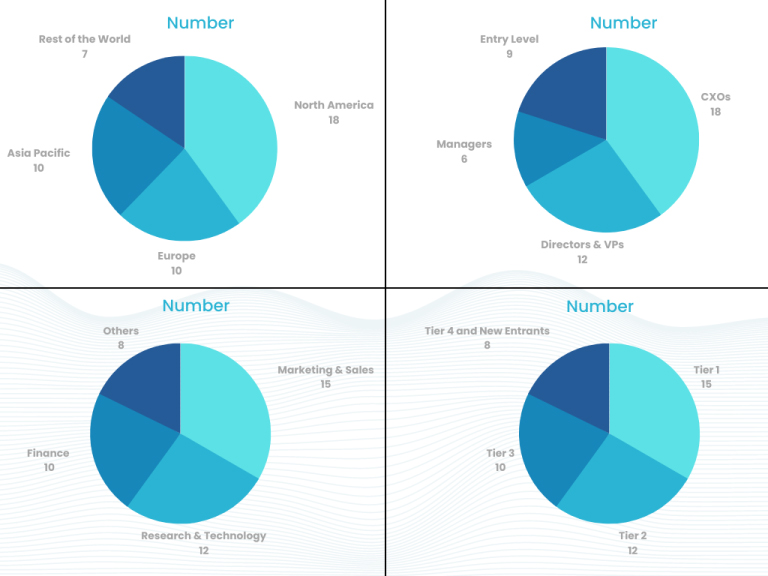
Part 1 wherein we interview the KOLs of major players as well as the upcoming ones across various geographic regions. This allows us to have their view over the market scenario and acts as an important tool to come closer to the accurate market numbers. As many as 45 paid and unpaid primary interviews are taken from both the demand and supply side of the industry to make sure we land at an accurate judgement and analysis of the market.
This step involves the triangulation of data wherein our team analyses the interview transcripts, online survey responses and observation of on filed participants. The below mentioned chart should give a better understanding of the part 1 of the primary interview.
Part 2: In this part of primary research the data collected via secondary research and the part 1 of the primary research is validated with the interviews from individual consultants and subject matter experts.
Consultants are those set of people who have at least 12 years of experience and expertise within the industry whereas Subject Matter Experts are those with at least 15 years of experience behind their back within the same space. The data with the help of two main processes i.e., FGDs (Focused Group Discussions) and IDs (Individual Discussions). This gives us a 3rd party nonbiased primary view of the market scenario making it a more dependable one while collation of the data pointers.
Step 3: Data Bank Validation
Once all the information is collected via primary and secondary sources, we run that information for data validation. At our intelligence centre our research heads track a lot of information related to the market which includes the quarterly reports, the daily stock prices, and other relevant information. Our data bank server gets updated every fortnight and that is how the information which we collected using our primary and secondary information is revalidated in real time.

Step 4: QA/QC Process
After all the data collection and validation our team does a final level of quality check and quality assurance to get rid of any unwanted or undesired mistakes. This might include but not limited to getting rid of the any typos, duplication of numbers or missing of any important information. The people involved in this process include technical content writers, research heads and graphics people. Once this process is completed the title gets uploader on our platform for our clients to read it.
Step 5: Final QC/QA Process:
This is the last process and comes when the client has ordered the study. In this process a final QA/QC is done before the study is emailed to the client. Since we believe in giving our clients a good experience of our research studies, therefore, to make sure that we do not lack at our end in any way humanly possible we do a final round of quality check and then dispatch the study to the client.
Key Segments
By Type
CK
Myoglobin
Lactate
WBC
Urea
CRP
Lipid and Protein Hydroperoxides
IL-6
Others
Request for Segment Customization as per your Business Requirement: Segment Customization Request
Regional Coverage:
North America
US
Canada
Mexico
Europe
Eastern Europe
Poland
Romania
Hungary
Turkey
Rest of Eastern Europe
Western Europe
Germany
France
UK
Italy
Spain
Netherlands
Switzerland
Austria
Rest of Western Europe
Asia Pacific
China
India
Japan
South Korea
Vietnam
Singapore
Australia
Rest of Asia Pacific
Middle East & Africa
Middle East
UAE
Egypt
Saudi Arabia
Qatar
Rest of Middle East
Africa
Nigeria
South Africa
Rest of Africa
Latin America
Brazil
Argentina
Colombia
Rest of Latin America
Request for Country Level Research Report: Country Level Customization Request
Available Customization
With the given market data, SNS Insider offers customization as per the company’s specific needs. The following customization options are available for the report:
Detailed Volume Analysis
Criss-Cross segment analysis (e.g. Product X Application)
Competitive Product Benchmarking
Geographic Analysis
Additional countries in any of the regions
Customized Data Representation
Detailed analysis and profiling of additional market players
The Wound Care Devices Market Size was valued at USD 2.6 Billion in 2023 and is expected to reach USD 4.0 Billion by 2032, and grow at a CAGR of 4.9%.
Burial Insurance Market was valued at USD 280 billion in 2023, projected to reach USD 463.7 billion by 2032, growing at a CAGR of 5.8% from 2024-2032.
The Surgical Robots Market Size was valued at USD 3.95 billion in 2023, and is expected to reach USD 8.79 billion by 2032, and grow at a CAGR of 9.3% over the forecast period 2024-2032.
The Venous Thromboembolism Treatment Market Size was valued at USD 1.05 Bn in 2023, and is expected to reach USD 2.13 Bn by 2032, and grow at a CAGR of 8.50% Over the Forecast Period of 2024-2032.
Isothermal Nucleic Acid Amplification Technology Market was valued at USD 4.68 billion in 2023 and is expected to reach USD 13.57 billion by 2032, growing at a CAGR of 12.40% from 2024-2032.
The Medical Billing Market was valued at USD 15.43 billion in 2023 and is expected to reach USD 41.32 billion by 2032, growing at a CAGR of 11.58% over the forecast period 2024-2032.
Hi! Click one of our member below to chat on Phone